
24 Aug 4 biotech Stocks Facing FDA rejection!
Clinical-Stage companies offer an exciting investment opportunity with massive upside potential. Most of these companies bring new and highly differentiated approaches, advanced scientific knowledge and a zeal for innovation to the table.
However, clinical trials are fraught with risk and uncertainty. Even the slightest setback can prove detrimental to the existence of these companies. Failure to meet clinical endpoints, lack of funding or rejection from regulatory authorities are risks that these companies have to bear in pursuit of excellence.
We take a look at some companies that have faced such setbacks in recent times:
Sesen Bio Inc. (NASDAQ: SESN)
Market Cap: $221.48M; Current Share Price: 1.13 USD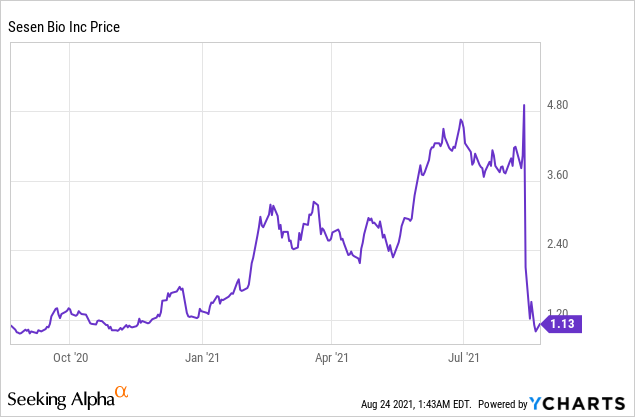
Data by YCharts
In August 2021, Sesen Bio received a Complete Response Letter from the FDA for its lead drug candidate Vicineum, which stated that the drug could not be approved “in its present form”. The FDA has asked for additional data and the Company intends to request for a Type A meeting, to improve its chances for approval. The Company had a late-stage meeting with the FDA in July, 2021 during which no Advisory Committee meetings or requests for post-marketing confirmatory trials were identified.
The Company is now facing a class action lawsuit for securities law violation amid reports that its 130-patient study had more than 2000 violations, out of which 215 were categorized as major, including complaints of “serious noncompliance” on the part of three investigators, which put “subjects at risk of harm”.
Sesen Bio is focused on creating targeted fusion protein therapeutics (TFPTs) for treatment of cancers. The Company was looking at a major catalyst in 2021, the approval of Vicineum, a potential best-in-class therapeutic for the treatment of non-muscle invasive bladder cancer (NMIBC) in the U.S. Sesen has submitted the MAA for Vicineum to the EMA in March 2021, and anticipates an EU approval in 2022.
The Company had submitted a completed Biologics License Application (BLA) to the FDA, supported by data from Phase 3 VISTA trial and positive chemistry, manufacturing and controls (CMC) data. The Company has requested for a priority review and was expecting a potential approval in mid-2021. Sesen was estimating peak revenue of $1B-$3B globally and $400M-$900M in the U.S.
Vicineum, a targeted fusion protein composed of an anti-EPCAM antibody fragment tethered to a truncated form of Pseudomonas Exotoxin A, which is capable of targeting cancer cells, with minimal peripheral toxicity. The candidate successfully completed a phase 3 VISTA Trial for treatment of non-muscle invasive bladder cancer (NMIBC) in patients who already underwent treatment with bacillus Calmette-Guérin (BCG), the current standard of care.
In May 2019, the Company released updated, preliminary primary and additional secondary endpoint data which demonstrated Vicineums’s clinically meaningful efficacy and favorable safety and tolerability. In December 2019, Sesen submitted a BLA submission for Vicineum to the FDA under rolling review.
Vicineum has a unique dual Mechanism of Action (MOA) that is a highly differentiated offering from existing treatment options. Its small size enables tumor penetration and far improved drug delivery. In January 2021, the Company released an independent analysis by ICER, a health technology assessment body that found Vicineum superior to the best supportive care, and showed Complete Response and Recurrence-Free Survival rates better than expected. In addition, the treatment also resulted in a decrease in cumulative health care costs of $101,000 by year five as compared to standard care options.
The Company’s pipeline is evaluating potential candidates for the treatment of additional cancers such as squamous cell carcinoma of the head and neck (SCCHN). Results from a phase 1 trial of an injectable form of Vicineum show anti-tumor activity and safety. In addition, patients demonstrated responses in non-injected tumors as well, implying vicineum’s ability to promote anti-tumor response and compatibility with other immunotherapies. Sesen is also developing deBouganin (VB6-845d), a highly potent plant toxin that can be de-immunized for systemic delivery.
FibroGen, Inc (NASDAQ: FGEN)
Market Cap: $ 1.131B; Current Share Price: 12.21 USD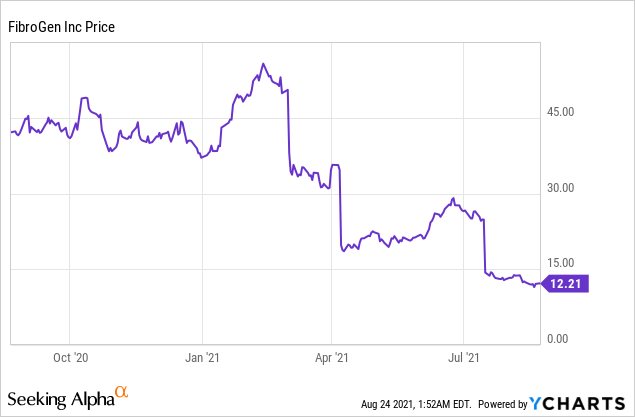
Data by YCharts
The Company faced a setback in August,2021, when the FDA declined to approve Roxadustat, intended for the treatment of anemia of chronic kidney disease, for want of more clinical data. A panel of experts, convened by the FDA, voted against the drug, with the FDA expressing concerns about the safety and efficacy of the drug previously.
FibroGen is a biopharmaceutical Company leveraging its expertise in hypoxia-inducible factor (HIF) and connective tissue growth factor (CTGF) biology to create a diverse pipeline of first-in-class therapeutics. The Company had a rough April, with shares of the company crashing on the heels of the disclosure that the safety analyses from a late-stage study of roxadustat in treating anemia of chronic kidney disease (CKD) included post-hoc changes to the stratification factors. The information essentially means that it can no longer claim that it is better at reducing major adverse cardiovascular events (MACE) when compared to erythropoiesis-stimulating agent epoetin alfa.
Roxadustat, the Company’s lead candidate, is a first-in-class, oral small molecule intended for the treatment of anemia associated with chronic kidney disease (CKD. The hypoxia-inducible factor prolyl hydroxylase (HIF-PH) inhibitor has demonstrated the ability to improve iron availability and reduce hepcidin. The product candidate has already been approved in China, Japan, and Chile for the treatment of anemia in CKD patients on dialysis and patients not on dialysis.
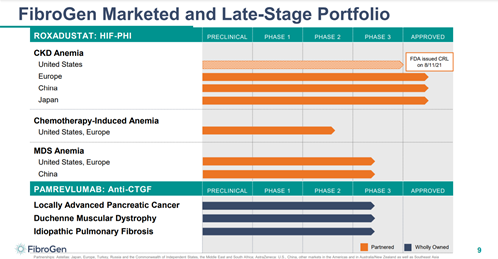
Image Source: Company
Astellas, its development partner, had filed the Marketing Authorization Application for roxadustat in Europe, which was accepted by the European Medicines Agency for review in May 2020. The drug was approved by the European Commission in August, 2021 and will be sold under the brand name Evrenzo. The sales from the drug are expected to bring in $690 million in Europe and Japan and the Company is eligible to receive a 22% royalty rate from Astellas, which could translate to $152 million as per analyst estimates.
In addition, it is also being evaluated in a Phase 2/3 development in China for anemia associated with myelodysplastic syndromes (MDS) and a Phase 2 U.S. trial for treatment of chemotherapy-induced anemia (CIA).
Furthermore, the Company is also developing Pamrevlumab, a fully-human monoclonal antibody, which is currently being evaluated in phase 3 clinical trials for the treatment of locally advanced unresectable pancreatic cancer (LAPC), Duchenne muscular dystrophy (DMD), and idiopathic pulmonary fibrosis (IPF). The candidate has been granted Orphan Drug Designation for LAPC, DMD, and IPF and Fast Track designation for LAPC and IPF. The candidate was recently awarded Rare Pediatric Disease Designation by the FDA for the treatment of Duchenne muscular dystrophy (DMD). The candidate is currently being evaluated in two Phase 3 trials in DMD.
The Company has had strategic collaboration with Astellas for development of hypoxia-inducible factor (HIF) anemia programs since 2004 for Japan, Europe, the Middle East, the Commonwealth of Independent States, and South Africa. FibroGen has entered into a partnership with AstraZeneca to develop the anemia programs in the U.S and other markets not licensed to Astellas, including China.
Upcoming catalysts for the company include Phase 2 topline data from the WHITNEY chemotherapy-induced anemia trial, which is expected to be released in the 3Q,2021. The Company also intends to announce the results from a Phase 3 anemia associated with MDS (MATTERHORN) trial in the second half of 2022 or the first half of 2023.
Spectrum Pharmaceuticals, Inc. (NASDAQ: SPPI)
Market Cap: $ 383.93M; Current Share Price: 2.34 USD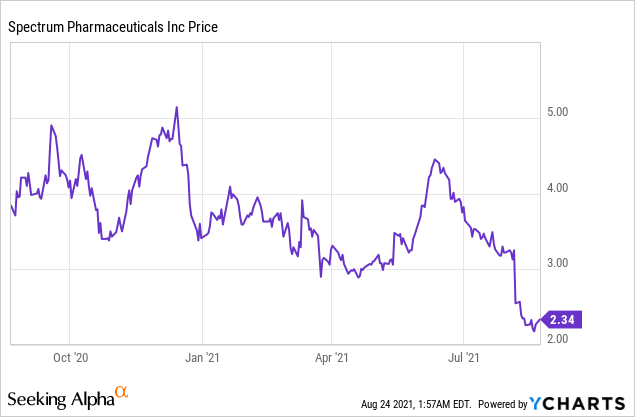
Data by YCharts
Spectrum Pharmaceuticals, a biopharmaceutical company, received a Complete Response Letter (CRL) for the Biologics License Application (BLA) for ROLONTIS® (eflapegrastim). The CRL asked for a reinspection of the Company’s manufacturing facilities to address deficiencies in manufacturing. The Company is seeking further clarification from the FDA and seeking to gain a better understanding of the “remediation timelines for the program”.
ROLONTIS® (eflapegrastim) is a long-acting granulocyte colony-stimulating factor (G-CSF) for the treatment of neutropenia in patients receiving myelosuppressive anti-cancer drugs. The candidate has been evaluated in two phase 3 trials namely ADVANCE and RECOVER, and has demonstrated non-inferiority (NI) in duration of severe neutropenia (DSN) and similar safety profile to pegfilgrastim. The Company’s BLA application was deferred by the FDA in October 2020, as COVID-19 pandemic travel restrictions derailed its inspection plan of the Hanmi Bioplant in South Korea, ROLONTIS® (eflapegrastim) manufacturing site.
The Company’s poziotinib, an orally administered irreversible tyrosine kinase inhibitor that targets EGFR and HER2 mutations, intended for the treatment of non-small cell lung cancer (NSCLC) in previously treated patients with HER2 exon 20 mutations, was granted a fast-track designation by the FDA in March 2021, Spectrum is planning a new drug application later this year. The Company presented data at the European Society for Medical Oncology Targeted Anticancer Therapies (ESMO TAT) Virtual Congress 2021, which showed that twice daily dosing (BID) improved anti-tumor activity and reduced toxicity compared to once daily dosing.
Refractory Non-Hodgkin Lymphoma (NHL)
Image Source: Company
In addition, the Company is also developing IGN002, an Interferon/ CD20 Monoclonal Antibody Fusion Protein that is targeting the treatment of Refractory Non-Hodgkin Lymphoma (NHL), and is currently being evaluated in a Phase 1 clinical trial.
Ardelyx Inc (NASDAQ: ARDX)
Market Cap: $ 148.49M; Current Share Price: 1.44 USD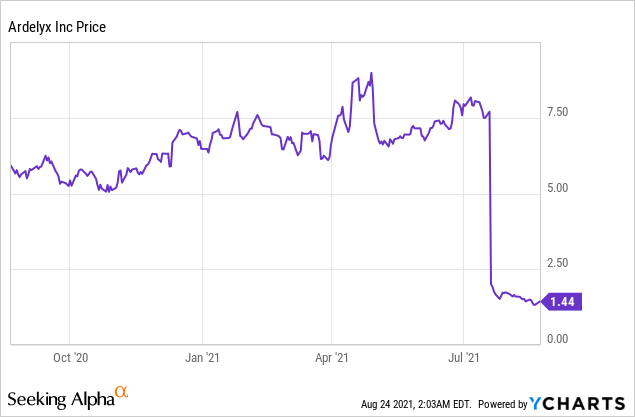
Data by YCharts
Ardelyx’s lead candidate Tenapanor, is a targeted, first-in-class, oral, small molecule intended for the control of serum phosphorus in adult patients with chronic kidney disease (CKD) on dialysis. The drug has the potential to become a foundational therapy for hyperphosphatemia and enable effective control of elevated phosphate in the blood. Tenapanor’s unique mechanism of action inhibits the sodium/hydrogen exchanger 3 (NHE3), by acting locally in the gut, resulting in decreased phosphate absorption through its effect on the paracellular pathway.
However, In July,2021 the Company received a Complete Response Letter (CRL) related to the New Drug Application (NDA) for tenapanor. The CRL states that the magnitude of the treatment effect as “small and of unclear clinical significance.” The FDA has requested Ardelyx to conduct an additional well-controlled trial that can demonstrate significant treatment effects of serum phosphorus. The Company intends to request for a Type A meeting with the FDA to determine the future course of action for the approval of tenapanor.
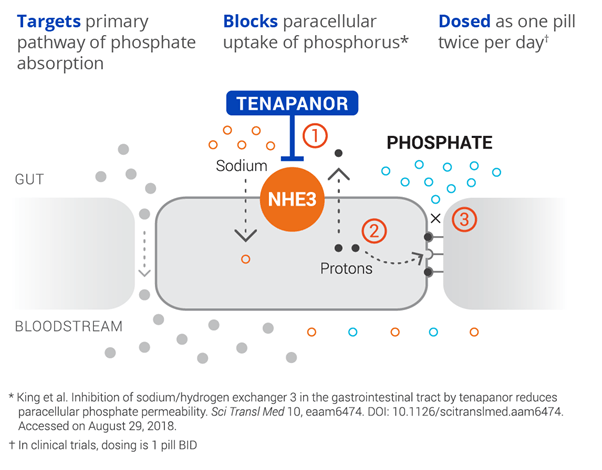
Image Source: Company
The Company has conducted three Phase 3 clinical trials, two monotherapy trials (BLOCK and PHREEDOM) and one dual mechanism trial (AMPLIFY), which demonstrate that Tenapanor can reduce phosphorus if taken twice a day, as both a monotherapy and as part of a dual-mechanism approach. The Company submitted a New Drug Application to the U.S. FDA in June, 2020 and was granted a Prescription Drug User Fee Act (PDUFA) of April 29, 2021 with a potential approval and eventual launch by Q3,2021.
In November 2017, Ardelyx entered into a licensing agreement with Kyowa Kirin for development and commercialization of Tenapanor for cardiorenal diseases and hyperphosphatemia in the Japanese Market. Furthermore, the Company has a licensing agreement with Shanghai Fosun Pharmaceutical Industrial Development Company Limited for development and commercialization of tenapanor for IBS-C and hyperphosphatemia related to chronic kidney disease in China. The Company has also entered into a licensing agreement with Knight Therapeutics, Inc. for commercial rights to tenapanor for the treatment of IBS-C and hyperphosphatemia in the Canadian market.
The Company’s unique approach is based on recreating and understanding unexploited ion transport mechanisms that are present in the gastrointestinal tract and kidney. Ardelyx is developing a pipeline of candidates that are orally active with minimal systemic absorption that reduce side-effects and drug-induced interactions. Ardelyx is developing RDX013 for the treatment of hyperkalemia that targets potassium secretion through the lumen of the gut and aims to lower the levels of serum potassium. The Company is also exploring other potential indications that could benefit from its unique approach.
Disclosure: I/we have no positions in any stocks mentioned, and no plans to initiate any positions within the next 72 hours.
Click here to please visit our detailed disclosure
References
https://fibrogen.gcs-web.com/static-files/6619528d-fa32-4c8f-b839-3373d2eda56a
https://finance.yahoo.com/news/spectrum-pharmaceuticals-receives-complete-response-113000081.html
https://investor.sppirx.com/index.php/static-files/2977ecaa-f5d9-42e3-91af-ed6738a39993
https://investor.sppirx.com/index.php/events-and-presentations





No Comments