
14 Jan 4 Promising Biotech Stocks with Immense Potential!
Clinical-Stage companies offer an exciting investment opportunity with massive upside potential. Most of these companies bring new and highly differentiated approaches, advanced scientific knowledge and a zeal for innovation to the table.
The Companies we have shortlisted this week have entered into strategic collaborations with some major pharmaceutical companies, had a successful drug launch and have upcoming catalysts in the form of data readouts, initiation of clinical trials and IND submission to look forward to.
However, a word of caution is in order as clinical trials are fraught with risk and uncertainty. Even the slightest setback can prove detrimental to the existence of these companies. Failure to meet clinical endpoints, lack of funding or rejection from regulatory authorities are risks that these companies have to bear in pursuit of excellence.
BioCryst Pharmaceuticals, Inc (NASDAQ: BCRX)
Market Cap: $2.91B; Current Share Price: 16.16 USD
Data by YCharts
BioCryst Pharmaceuticals released preliminary fourth-quarter revenue figures for its lead drug Orladeyo (berotralstat), a first-in-class oral therapy for treatment of hereditary angioedema (HAE) induced attacks in adults and paediatric patients 12 years and older and works by inhibiting the activity of plasma kallikrein. Currently the drugs used for the treatment of the condition are all intravenously administered, and the candidate promises improvement both in the ease of administration and cost of treatment.

Image Source: Company
Unaudited fourth-quarter revenue for the drug was $45.6 million during the last three months of 2021, an increase of 23% over the previous quarter, while unaudited net revenue for full year 2021 was $122 million. There is a steady new patient demand for ORLADEYO, with new patient additions remaining consistent through the previous four quarters, driven by patients switching from other prophylactic therapies and acute-only therapy.
The management also expects the revenue from Orladeyo to be no less than $250 million in 2022, reiterating its potential to reach $1 billion in annual sales. The Company also reported a patient retention rate of 70% through the first year of its launch. The drug is now covered by major payors as well as regional pharmacy benefit managers. According to market research including 60 U.S physicians, the drug usage is expected to double, with ORLADEYO becoming the most prescribed prophylactic treatment in the next 12 months.
In January 2021, the Company had reported the grant of a marketing and manufacturing approval for oral, once-daily ORLADEYO™ (berotralstat) 150 mg for prophylactic treatment of hereditary angioedema (HAE) in adults and paediatric patients 12 years and older in Japan.
Hereditary Angioedema is a genetic disorder that causes recurrent swelling in the skin, gastrointestinal tract and upper airway. The disorder is caused by mutation in the C1NH gene that regulates the C1 inhibitor protein, and is categorised into Type I, Type II and Type III and is marked by deficiency of C1 inhibitor and dysfunction of C1 inhibitor and unknown source respectively. According to a report by Industry Research the Hereditary Angioedema market is expected to reach 3.829 Billion USD in 2025, growing at a CAGR of 9.7% from 2.197 Billion USD in 2019.
BioCryst follows a structure-guided design approach that involves creating synthetic compounds to target active sites of proteins, whose dysfunction causes diseases. The Company uses a combination of traditional biology, medicinal chemistry and advanced techniques like x-ray crystallography, computer modelling of molecular structures, virtual screening, and protein biophysical chemistry to arrive at a clear idea of the three-dimensional structure of the active enzyme. The initial focus of the Company is on two rare diseases namely Hereditary angioedema and Fibrodysplasia ossificans progressiva (FOP).
BioCryst has an FDA approved antiviral drug Rapivab, which is used for the treatment of acute uncomplicated flu in patients who are two years and older. The Company has another drug in its pipeline, namely Galidesivir, which has demonstrated the potential to fight numerous families of viruses. The drug has successfully completed phase I clinical study and has passed safety tests in humans. The candidate was primarily developed for treating Ebola but will now be developed to focus on biodefense threats, such as Marburg virus disease according to the Company.
The Company’s pipeline consists of BCX9930, an oral Factor D inhibitor, intended for the treatment of complement-mediated diseases, BCX9250, an oral ALK-2 inhibitor for treatment of Fibrodysplasia Ossificans Progressiva (FOP) and Galdidesivir, a broad-spectrum antiviral for serious viral illnesses.
Century Therapeutics Inc (NASDAQ: IPSC)
Market Cap: $814.40M; Current Share Price: 14.38 USD
Data by YCharts
Century Therapeutics announced a research collaboration and license agreement with Bristol Myers Squibb (NYSE: BMY), to develop and commercialize up to four programs in acute myeloid leukemia and a program in multiple myeloma, with an option to include two additional programs subject to conditions. The Company will be leveraging its expertise in induced pluripotent stem cell (“iPSC”) derived, engineered natural killer cell (“iNK”) and / or T cell (“iT”) therapies to develop therapeutics for hematologic malignancies and solid tumors.
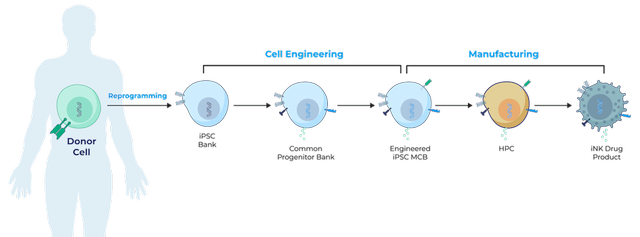
Image Source: Company
Century will be responsible for candidate discovery and preclinical development, while Bristol Myers Squibb will be responsible for clinical development and commercialization activities. The Company will receive an upfront payment of $100 million, in addition to $50 million equity investment in the Company’s stock at a price of $23.14 per share. Furthermore, Century is eligible to receive development, regulatory, and commercial milestone payments totaling more than $3 billion across the four programs.
The Company’s cell therapy platform can generate allogeneic, iPSC-derived NK and T cell products, by using its expertise in induced pluripotent stem cells (iPSCs), gene editing, protein engineering and manufacturing. Century’s core competencies include efficient precision gene editing technology, proprietary Allo-Evasion technology, CAR and protein engineering, common engineered IPSC Progenitors and Streamlined Manufacturing.
Century is developing a robust portfolio of candidates that utilize its Allo-Evasion™ technology, which enables product candidates to avoid host rejection and increase the durability of clinical responses.
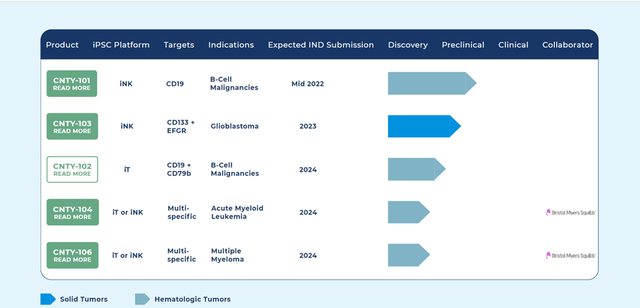
Image Source: Company
The Company’s lead candidate is CNTY-101, an allogeneic, iPSC-derived CAR-iNK cell therapy, intended for the treatment of relapsed, refractory B-cell lymphoma. The Company is aiming to file an IND for the candidate by mid-2022. The pipeline also consists of CNTY-103, a CAR-iNK candidate that targets glioblastoma and is the Company’s first solid tumor candidate. Some of the other candidates in the pipeline include CNTY-104 and CNTY-106 that target Acute Myeloid Leukemia and Multiple Myeloma, which are now being developed in collaboration with BMS.
CareDx Inc (NASDAQ: CDNA)
Market Cap: $2.15B; Current Share Price: 40.74 USD
Data by YCharts
CareDx, a precision medicine solutions Company aims to discover and develop differentiated solutions for transplant patients and caregivers. The Company offers testing products, services, digital healthcare solutions and genomics-based information for transplant patients through their entire transplant journey. The Company is a leading partner of major US transplant centres and has forged extensive clinical partnerships across the transplant network.
The Company’s product offerings encompass transplant lab services that include AlloSeq cfDNA, a kit-based blood test solution for precise management of cfDNA and AlloSeq Tx17 that provides superior HLA typing using an innovative Hybrid Capture Technology among others. CareDx also provides digital transplant solutions such as Ottr Organ, Ottr Cellular, XynQAPI to name a few. The Company also offers pharma services to biopharmaceutical companies to run multicenter clinical trials through products and services like the highly sensitive NGS surveillance for allogeneic cell therapy.

Image Source: Company
The Company published results from ADMIRAL (Assessing AlloSure Dd-cfDNA Monitoring Insights of Renal Allografts with Longitudinal Surveillance) study, a multicenter prospective study that evaluated AlloSure® Kidney as an accurate measure of allograft injury in routine organ transplant surveillance.
The study monitored 1,092 kidney transplant patients from 7 transplant centers for up to 3 years using the AlloSure Kidney donor-derived cell-free DNA (dd-cfDNA) as part of the standard of care and demonstrated that AlloSure® Kidney strongly correlated with clinical and subclinical allograft rejection (p<0.001), is associated with a 271% increased risk of dnDSA development (p=0.001), was elevated 91 days (median) ahead of DSA identification, showed a 62% relative improvement over serum creatinine in identifying subclinical and clinical rejection and predicted a > 25% decline in eGFR over 3 years (HR 1.97, p = 0.041), while persistently low levels identify allograft quiescence.
As per its Q3,2021 financial results, the Company reported an 86% growth YoY in patient test results, a 42% YoY revenue growth in 2021, and a greater than $300M Annual Revenue Run Rate. The Company is strategically investing in clinical development through large multicenter clinical studies evaluating long-term transplant outcomes in COVID, Kidney and Heart. In addition, CareDX is also engaged in multiple investigator-initiated trial programs in over 40 transplant centers such as Cedars Sinai, Stanford Medicine, John Hopkins and Columbia University Medical Center among others.
Furthermore, the Company is also partnering with leading cell therapy biopharmaceutical companies
Dyadic International, Inc (NASDAQ: DYAI)
Market Cap: $108.67M; Current Share Price: 3.89 USD
Data by YCharts
In December 2021, Dyadic entered into a research and license collaboration agreement with Janssen Biotech Inc. The Company will receive an upfront payment of $500,000 in exchange of non-exclusive rights to its C platform that can develop C1 production cell lines for manufacturing Janssen’s therapeutic protein candidates. Janssen will bear the research and development costs associated with developing the C1 production cell lines for its product candidates and has an option to pay a mid-seven figure for an exclusive license from Dyadic for manufacturing therapeutic proteins directed to one specific target.
The Company is eligible to receive development and regulatory milestones in the mid-seven figures for each of the product candidates, in addition to commercial milestone payments in the low nine figures per product, subject to a limit. The total amount will depend on the cumulative amount of active pharmaceutical ingredient produced using Dyadic’s C1 platform in each of the product candidates.
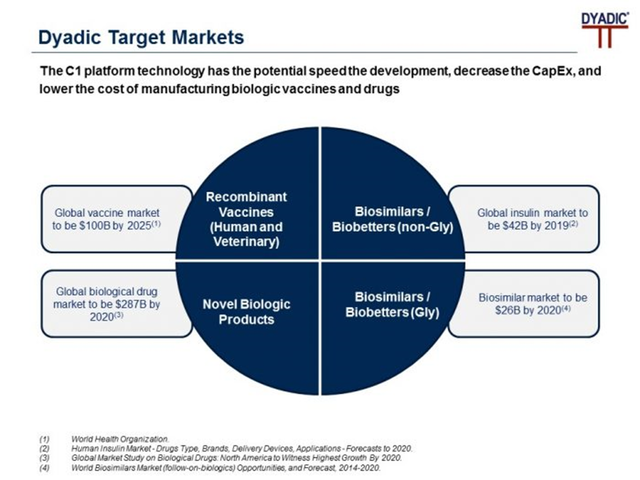
Image Source: Company
Dyadic is leveraging its patented C1 technology platform, a premier gene expression system, which has the potential to be an efficient expression system for developing and manufacturing biologic vaccines and drugs by bio pharmaceutical companies. The platform can be used to change the morphology of an organism resulting in better growth conditions and productivity and facilitates the production of purer enzymes and other proteins through C1 White StrainTM. The Company is actively seeking partners for sub-licensing its C1 platform technology in the vaccine, antibody and biosimilar industries.
The technology has wide application in industries such as biobetters, biopharmaceuticals, enzyme therapies, animal health and enzyme catalysts among others.
The Company sold its industrial technology business to DuPont in December 2015 and retained co-exclusive rights to the C1 technology for use in human and animal pharmaceutical applications. Dyadic can enter into sub-license agreements subject to certain exceptions. Currently, the Company is working on COVID-19 related vaccines, including development of DYAI-100, its proprietary COVID-19 vaccine candidate, which will be evaluated in a first-in-human Phase 1 clinical trial.
Disclosure: I/we have no positions in any stocks mentioned, and no plans to initiate any positions within the next 72 hours.
Click here to please visit our detailed disclosure
References
https://www.centurytx.com/science/
https://www.centurytx.com/pipeline/
https://caredx.com/products-and-services/transplant-services/
https://investors.caredxinc.com/static-files/11fe8cce-ceaa-412c-b945-0febba072cbd
https://finance.yahoo.com/news/caredx-partners-multiple-biotechnology-companies-120000627.html





No Comments