
18 May 5 Biotech Stocks with Upcoming Catalysts!
Biotech Companies continue to show resurgence even in these challenging times, with more and more companies persevering with bringing their clinical candidates to fruition. While Vaccine development and therapeutics to combat the COVID-19 pandemic are at the forefront of development activities in the biotech sector, there are some companies that are making strides in other related fields such as neurodegenerative diseases and oncology. We bring to you some companies that are leveraging their leading-edge technology to make innovative therapeutics in areas that have a large unmet need.
Vanda Pharmaceuticals, Inc. (NASDAQ: VNDA)
Market Cap: $1.03B; Current Share Price: 18.58 USD
Data by YCharts
Vanda has two products in the market namely Hetlioz (tasimeltion) and Fanapt (iloperidone), intended for the treatment of Non-24-Hour Sleep-Wake Disorder in adults/Nighttime sleep disturbances in Smith-Magenis Syndrome (SMS) and schizophrenia in adults respectively. In December 2020, the U.S. Food and Drug Administration (FDA) approved HETLIOZ® capsule and liquid formulations for the above-mentioned indications.
The Company reported total net product sales from HETLIOZ® and Fanapt®, which stood at $62.7 million in the first quarter of 2021,an increase of 8 percent, as compared to $58.0 million in the first quarter of 2020. The net product sales from HETLIOZ® were $39.3 in Q1,2021 as against $35.3 million Q1,2020, an increase of 11 percent.
The Company is leveraging its expertise in genetics and genomics to deliver novel medicine to patients in areas with large unmet medical needs.
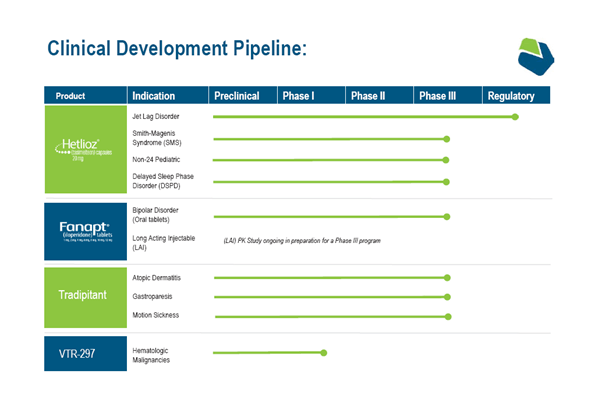
Image Source: Company
The Company’s pipeline consists of Hetlioz, which is currently being evaluated in a Phase III trial in n delayed sleep phase disorder (DSPD) and is currently enrolling patients in a Phase III clinical study of tradipitant in gastroparesis. Vanda is targeting the completion of enrollment of around 200 patients for the study by Q2,2021 and intends to file a New Drug Application (NDA) in late 2021 or early 2022.
Vanda had to pause a Phase III clinical trial of Fanapt® in bipolar disorder in 2020 due to the ongoing COVID-19 pandemic and resumed it during Q1,2021. The Company has also initiated the development of Fanapt® in Parkinson’s disease psychosis (PDP) in Q1,2021. The Company is also pursuing the development of Tradipitant in Atopic Dermatitis and Motion Sickness, besides conducting a phase 1 clinical study of VTR-297 for Hematologic Malignancies.
Epizyme Inc (NASDAQ: EPZM)
Market Cap: $918.80M; Current Share Price: 9.01 USD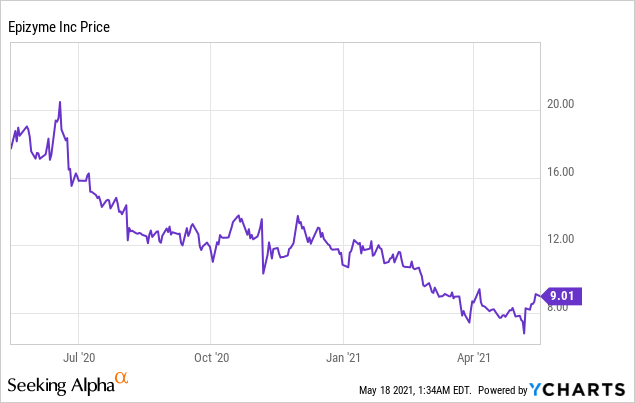
Data by YCharts
With Epigenetics at the core of its novel therapeutic inventions for treatment of cancer and other serious illnesses, the company is pioneering the development of small molecule inhibitors of histone methyltransferases (HMTs), histone acetyltransferases (HATs) and helicases. The Company is focused on creating oral medication that can target specific diseases and offer ease of administration and tolerability.
In January 2020, the FDA approved Tazverik, the very first EZH2 inhibitor to receive an approval, for treatment of adults and pediatric patients aged 16 years and above with metastatic or locally advanced epithelioid sarcoma not eligible for complete resection. This was followed by an approval for a supplemental new drug application for Tazverik (tazemetostat) for the treatment of follicular lymphoma (FL) indications. The Company is conducting a Phase 1b/3 Confirmatory trial in Follicular Lymphoma and Epithelioid Sarcoma.
Furthermore, the Company is also developing a PRMT 5 Inhibitor and PRMT 1 Inhibitor for the treatment of Solid Tumors and blood cancers, in collaboration with GlaxoSmithKline. GSK holds the global development and commercialization rights to the candidate.
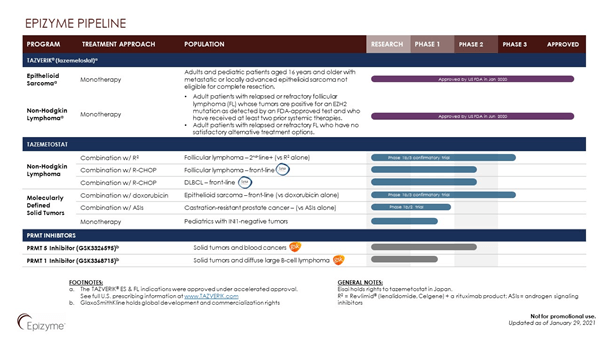
Image Source: Company
Epizyme has entered into key strategic collaborations with companies such Roche Group, Lymphoma Study Association (LYSA), EISAI and GSK for advancing the development of its drug candidates as well as exploring a range of research and development programs. As part of its arrangements with EISAI, it has retained the global rights to tazemetostat, an EZH2 inhibitor, outside of Japan for an upfront payment of $40 million and consideration of future royalties.
In December 2012, the Company entered into an agreement with Roche Molecular Systems, Inc., to develop a diagnostic to identify patients who have certain EZH2 mutations, which can be targeted by tazemetostat. The Company is currently engaged in a Phase 2 study in Non-Hodgkin lymphoma (NHL).
LYSA and its collaboration are for the investigation of its tazemetostat with R-CHOP, a standard chemotherapy for NHL, as a front-line treatment in elderly, high-risk patients with newly diagnosed DLBCL.
The Company has entered into a collaboration and licensing agreement with GSK for development of novel small molecule HMT inhibitors. GSK has paid an upfront payment of $20 million, with research, development and regulatory milestone payments of up to $402 million and sales-based milestone payments of up to $218 million in the offing for its novel small molecule HMT inhibitors.
Epizyme has been generating a lot of interest on the potential of Tazverik in the treatment of follicular lymphoma, where it can be used for both treatment and as maintenance therapy, which analysts believe provides “significant upside to current valuation”.
As per its Q1,2021 financial results, TAZVERIK’s net product revenue from Epithelioid Sarcoma (ES) and Follicular Lymphoma stood at $6.2 million in Q1,2021, the best demand since launch and an increase of 37%. New prescribing accounts also grew by 38 percent compared to Q4,2020, in spite of the effect of Covid-19 pandemic on physician visits by cancer patients.
The Company is currently evaluating Tazemetostat in Follicular Lymphoma, prostate cancer and conducting confirmatory Phase 1b/3 Study of TAZVERIK in Epithelioid Sarcoma. In addition, the candidate is undergoing Basket Trials in additional Hematologic Malignancies and Solid Tumors. Epizyme is planning to submit an Investigational New Drug (IND) application for its SETD2 inhibition in indications such as multiple myeloma and in other B-cell malignancies such as large-cell lymphoma in the mid-2021.
Market Cap: $966.91M; Current Share Price: 5.78 USD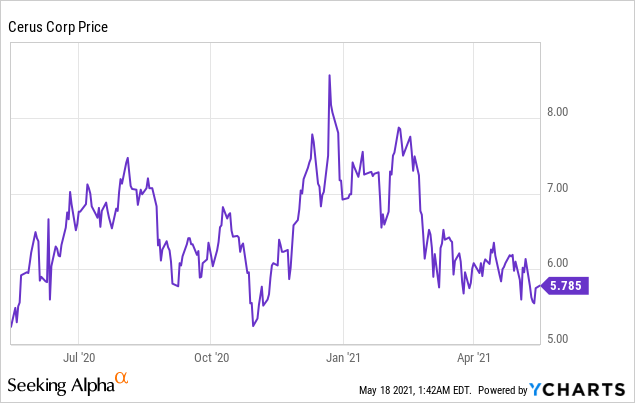
Data by YCharts
Cerus aims to become the preeminent global blood products company through its proprietary INTERCEPT Blood System. The Company is developing and supplying cutting-edge technologies and pathogen-protected blood components, to hospital and blood suppliers worldwide. Cerus offers its products and services in the United States, Canada, Europe, the Middle East, Asia, Africa and Latin America.
The Company’s Products consists of the Intercept Blood System – Platelets, which has been commercialized in the U.S as well as other major geographies. The Company is refiling for a CE mark under the Medical Device Regulation commencing in 2021. In addition, a post- post-approval haemovigilance study is currently enrolling patients in the U.S.
Cerus was granted an FDA approval of the premarket approval supplement (PMA) to produce PRCFC for its INTERCEPT Blood System—Plasma. The Company is currently engaged in two crucial Phase 3 clinical trials, known as the RedeS and ReCePI studies and is enrolling patients in the U.S. There are plans to initiate additional studies in the U.S, besides preparing a submission for its INTERCEPT Blood System—Red Blood Cells in Europe in 2021. Furthermore, the Cerus received an FDA approval for INTERCEPT Blood System—Cryoprecipitation in November 2020 and is in preparation for a limited launch in the U.S in 2021, followed by national commercialization in 2022.
Cerus has landed a contract with Canadian Blood Services, one of the two blood operators in the country, which caters to all provinces and territories except Quebec, for provision of pathogen inactivated platelets in Canada. The services will be gradually implemented in the Ottawa region in Fall 2021, based on approval from Health Canada for extension of pathogen reduced platelets’ shelf life from five days to seven, after which the company is looking at broader implementation.
Passage Bio (NASDAQ: PASG)
Market Cap: $836.65M; Current Share Price: 14.82 USD
Data by YCharts
Passage Bio’s portfolio consists of three lead candidates for the treatment of GM1 Gangliosidosis, Frontotemporal dementia (FTD) and Krabbe Disease. PBGM01, the Company’s gene therapy product candidate, uses an AAVhu68 viral vector to deliver functional β-gal enzyme, which is delivered directly to the cerebrospinal fluid by an intra-cisterna magna (ICM) injection. Preclinical studies demonstrate that a single-dose of the candidate provided multiple benefits such as restoring β-gal activity in the brain and peripheral tissues, prevented onset of neurological signs, reduced brain lesions and improved survival.
The candidate is currently being evaluated in a multinational Phase 1 / 2 (Imagine-1) to study the safety, tolerability and efficacy of PBGM01 in patients with Early Onset Infantile (Type 1) and Late Onset Infantile (Type 2a) GM1. In addition, the Company is also developing PBFT02, a gene therapy, which uses an AAV1 viral vector to deliver granulin (GRN) gene for the treatment of FTD-GRN, which is currently undergoing phase 1/2 trial namely upliFT-D.

Image Source: Company
PBKR03, a next-generation AAVhu68 capsid designed to deliver DNA encoding the galactosylceramidase (GALC) enzyme for the treatment of infantile Krabbe disease, is currently under clinical development in a phase 1/2 trial named GALax-C.
Passage Bio is also targeting the treatment of Metachromatic Leukodystrophy through its candidate PBML04 and is conducting IND-enabling studies at present. Furthermore, the Company’s discovery pipeline consists of PBCM06 for the treatment of Charcot-Marie-Tooth Tyfpe 2A and an undisclosed candidate that is targeting CNS disorders.
The Company has entered into a collaboration with the University of Pennsylvania’s Gene Therapy Program (GTP), which gives it access to capsid engineering and next-generation capsid libraries, vector engineering, transgene design and gene therapy modalities, animal disease models and related studies for lead-optimization of product candidates. The agreement also provides the option to license 11 additional programs, with global commercial rights to the products and exclusive rights to certain technologies.
In May 2021, the Company entered into a collaboration with InformedDNA, to provide no-cost genetic counseling and testing for adults diagnosed with Frontotemporal Dementia (FTD).
The Company has multiple upcoming catalysts in the form of initial safety and 30-day biomarker data from Phase 1/2 PBGM01 trial in the fourth quarter of 2021, dosing of first patient in Phase 1/2 FTD-GRN trial in Q2 or Q3, 2021, followed by initial safety and 30-day biomarker data in the second half of 2022. In addition, the Company intends to dose the first patient in Phase 1/2 Krabbe trial in Q3,2021 and report initial safety and 30-day biomarker data in the second half of 2022.
Prothena Corporation, Plc (NASDAQ: PRTA)
Market Cap: $1.02B; Current Share Price: 23.00 USD
Data by YCharts
Prothena’s approach is based on targeting protein dysregulation and is being used to develop a pipeline that is focused on offering novel therapeutics for rare peripheral amyloid and neurodegenerative diseases. The Company is leveraging its expertise in neurological dysfunction and misfolded proteins such as Aβ, tau, alpha-synuclein and transthyretin to target pathogenic proteins that are responsible for various diseases.
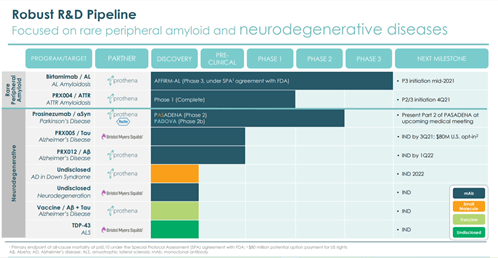
Image Source: Company
The Company has an active neurotherapeutics discovery program which is targeting the treatment of neurodegenerative diseases such as Alzheimer’s disease, frontotemporal dementia and amyotrophic lateral sclerosis (ALS). Prothena has entered into a collaboration with Bristol-Myers Squibb for development of PRX005, TDP-43 and an undisclosed target. Prothena’s neuroscience pipeline consists of programs that are currently under development for the treatment of Alzheimer’s disease including PRX012 that targets Aβ (Amyloid beta), birtamimab, intended for the treatment of AL amyloidosis and PRX004 for the potential treatment of ATTR amyloidosis.
Prothena is also collaborating with Roche for the development of prasinezumab, which targets alpha-synuclein and is intended for the treatment of Parkinson’s disease. The Company’s candidates are currently undergoing various clinical trials including a phase 3 study of Birtamibab, a Phase 2 study of Prasenizumab and a phase 1 study of PRX004.
The Company is poised to receive up to $405M in the next 5 years in the form of IND and Phase 1 based payments from its partner programs. Prothena is gearing up for a confirmatory phase 3 global study of birtamimab in mid-2021. The Company dosed the first patient in a Phase 2b PADOVA study in May 2021 and received $60 million in milestone payment from Roche.
Disclosure: I/we have no positions in any stocks mentioned, and no plans to initiate any positions within the next 72 hours.
Click here to please visit our detailed disclosure
References
https://www.epizyme.com/programs/pipeline/
https://www.biospace.com/article/epizyme-s-tazverik-picks-up-second-fda-approval-of-2020/
https://vandapharmaceuticalsinc.gcs-web.com/node/14506/pdf
https://www.vandapharma.com/investors?locale=en/#news
https://s26.q4cdn.com/467285195/files/doc_financials/2020/Cerus-2020-Annual-Report.pdf
https://finance.yahoo.com/news/cerus-corporation-announces-contract-canadian-130000131.html
https://ir.prothena.com/static-files/a4ea07aa-950d-4f21-bad9-3f8c02f1e127





No Comments