
02 Jun 5 Companies For Your Investment Watchlist!
Biotech Companies continue to show resurgence even in these challenging times, with more and more companies persevering with bringing their clinical candidates to fruition. While Vaccine development and therapeutics to combat the COVID-19 pandemic are at the forefront of development activities in the biotech sector, there are some companies that are making strides in other related fields such as immune-oncology, lung and cervical cancer. Some of these companies have entered into strategic collaborations with some major pharmaceutical companies and have upcoming catalysts in the form of IND submissions. We take a look at some of them below:
Pieris Pharmaceuticals, Inc (NASDAQ: PIRS)
Market Cap: $200.56M; Current Share Price: 3.44 USD
Data by YCharts
Pieris Pharmaceuticals recently entered into a multi-program research collaboration and license agreement with Genentech, to develop and commercialize respiratory and ophthalmology therapies, using Pieris’ proprietary Anticalin® technology. The agreement involves an upfront payment of $20 million to Pieris, along with eligibility to receive another $1.4 billion in additional milestone payments as well as tiered royalties for any program that reaches commercialization. Pieris is responsible for the discovery, research and pre-clinical development of any potential candidate, while Genentech will handle IND-enabling activities, clinical development, and commercialization. Genentech can choose additional targets for an option exercise fee.
The Company is developing PRS-060/AZD1402, an inhaled IL-4 receptor alpha inhibitor, intended for the treatment of moderate-to-severe asthma in collaboration with AstraZeneca. The candidate has initiated dosing in the first part of a global phase 2a study and expects to announce the result from the trial next year. In addition, Pieris is planning a two-arm phase 2 study for cinrebafusp alfa, a 4-1BB/HER2 bispecific for the treatment of HER2-expressing solid tumors, in gastric cancer, in collaboration with Lilly and Seagen. The first arm of the study will evaluate the combination of ramucirumab and paclitaxel in HER2-high gastric cancer, while the second arm will study the combination with tucatinib in HER2-low gastric cancer.

Image Source: Company
The Company develops Anticalin-based drugs for treatment of conditions related to immuno-oncology and respiratory diseases such as Asthma and Anaemia. Anticalin proteins are engineered versions of lipocalins that are human proteins having the ability to bind, store and transport molecules, which the Company has leveraged for delivering drug candidates binding to a broad spectrum of therapeutic targets, in the clinic and in partnerships with leading pharmaceutical companies. The technology was licensed from the Technische Universität München (TUM) and provides it exclusive access to Anticalin-related IP.
In 2017, the Company signed deals with Servier in immuno-oncology and is planning to present preclinical data for PRS-344/S095012, a 4-1BB/PD-L1 bispecific, at the AACR Annual Meeting 2021. The candidate will advance into phase 1 development this year. Pieris holds the exclusive commercialization rights in the U.S and is eligible for royalties on ex-us sales. Both companies are also developing PRS-352, an undisclosed bispecific. Pieris has a strong patent portfolio, comprising key patents granted in the United States, Europe and Asia, covering the Anticalin® protein as a drug class, libraries of Anticalin proteins and all derived products.
Syros Pharmaceuticals, Inc (NASDAQ: SYRS)
Market Cap: $398.99M; Current Share Price: 6.45 USD
Data by YCharts
The Company’s core area of interest is controlling and regulating gene expression and developing therapeutics that leverages its proprietary gene control platform. Syros uses its path breaking platform to identify disease-causing genes, and develop therapeutics to control them, thereby addressing the need for a cure for diseases which were hitherto side-lined for drug discovery and development, owing to a poor understanding of their gene expression.
Syros’s pipeline consists of SY-1425 an oral first-in-class selective retinoic acid receptor alpha (RARα) agonist, which is being evaluated in a phase 3 trial in RARA-positive patients with newly diagnosed higher-risk myelodysplastic syndrome (HR-MDS). The candidate is also being developed for the treatment of acute myeloid leukaemia (AML) and will undergo phase 2 clinical trials in 2H,2021.
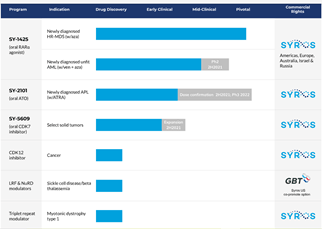
Image Source: Company
SY-5609, an oral inhibitor of the cyclin-dependent kinase 7 (CDK7) is currently engaged in a phase 1 dose-escalation studies for treatment of advanced breast, colorectal, lung, ovarian or pancreatic cancer, or with solid tumors of any histology that harbor Rb pathway alterations.
Syros is also developing SY-2101, an oral arsenic trioxide (ATO) intended for the treatment of newly diagnosed acute promyelocytic leukaemia (APL). The candidate is being advanced into a dose confirmation study and will consecutively undergo a phase 3 study in 2022.
Syros has a worldwide strategic collaboration and option agreement with Incyte Corporation, for discovering and developing up to seven novel therapeutic targets for myeloproliferative neoplasms (MPNs). The Company has also entered into a strategic collaboration with Global Blood Therapeutics (GBT) to identify and validate a novel fetal hemoglobin repressor through the use of its broader drug discovery effort in sickle cell disease and beta thalassemia.
22 Century Group Inc (NYSE: XXII)
Market Cap: $686.00M; Current Share Price: 4.50 USD
Data by YCharts

Image Source: Company
A leading plant-based biotechnology Company, 22 century group works on altering the nicotine levels in tobacco cigarettes by leveraging its expertise in genetic engineering. The Company’s cigarettes have 95 percent less nicotine as compared to conventional cigarettes and are developed using cutting-edge gene-editing and modern plant breeding techniques.
The Company is also using its proprietary technology to lower cannabinoid levels in hemp/cannabis plants and develop plants with valuable profiles and agronomic traits, which can be commercialized through its strategic partnerships in the industry.
22 Century group has built a robust intellectual property rights portfolio dealing with both tobacco plants and hemp/cannabis plants. The Company’s cutting-edge genetic engineering helps reduce the biosynthesis of nicotine and other alkaloids in the tobacco plant, without impacting its characteristics, taste and aroma. In addition, the technology can also be used to regulate and propagate specific content and profiles which may be considered desirable.
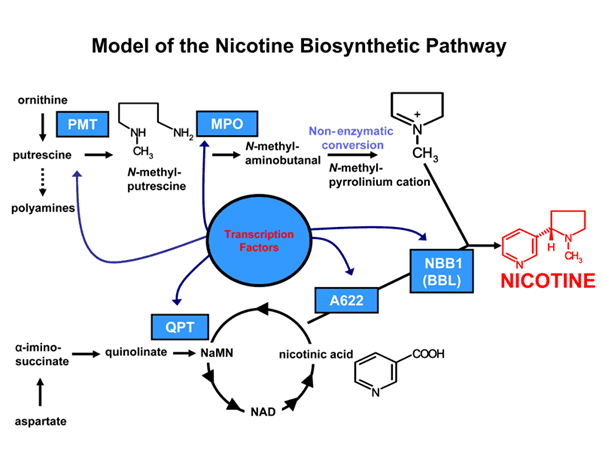
Image Source: Company
The Company has entered into a research and a worldwide license agreement with Anandia Laboratories Canada, for an exclusive sublicense to the patents and patent applications related to hemp/cannabis plant production in the United States and a co-exclusive sublicense for the rest of the world, except Canada. Furthermore, the Company has an ongoing collaboration with KeyGene, which provides access to genomics, molecular genetics, trait discovery and plant breeding technologies.
22 Century Group’s SPECTRUM® cigarettes were chosen by the National Institute on Drug Abuse (NIDA) for independent scientific clinical studies. The Company has delivered more than 28 million SPECTRUM® research cigarettes to over 100 independent scientific clinical studies.
Moonlight, the Company’s proprietary menthol cigarettes, were granted an FDA authorization in December 2019. This is the third product to market its products under the FDA’s PMTA regulatory pathway, besides products from Philip Morris International and Swedish Match.
VLN, a companion product to moonlight cigarettes, is currently under review by the FDA as the Company has submitted a Modified Risk Tobacco Product (MRTP) application for grant of a marketing authorization and permission to inform customers that these contain “95% Less Nicotine.”
The Company believes that it has the potential to target a 1.3 trillion market opportunity including tobacco, hemp/cannabis and a third plant-based franchise. The grant of an MRTP designation will enable the Company to enter into strategic distribution, international licensing, and partnership opportunities. 22 Century is fully prepared for the launch of VLN and is likely to initiate commercial sales within 90 days of the authorization. The Company has finished the groundwork for the launch including identification of initial launch markets, discussions with trade partners and preparing marketing material.
The Biden Administration is calling for change in federal policy concerning tobacco products including pushing for reduction in the amount of nicotine in all combustible cigarettes to be minimal or non-addictive. In addition, the government is working on eliminating the use of menthol in cigarettes. This augurs well for the Company and its products in the long-run.
Tyme Technologies (NASDAQ: TYME)
Market Cap: $262.46M; Current Share Price: 1.54 USD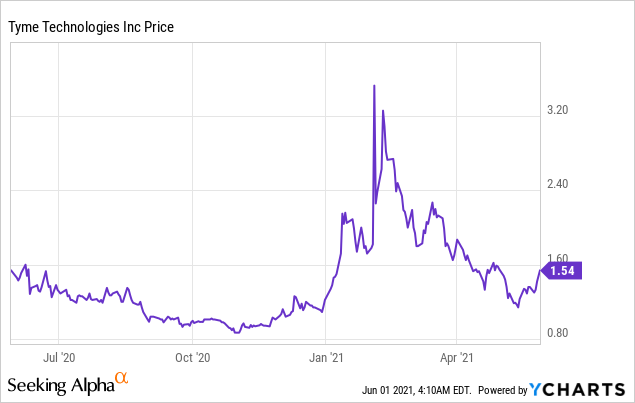
Data by YCharts
Tyme is a clinical-stage biotechnology company that is developing cancer metabolism-based therapies for metastatic and hard-to-treat cancers with large unmet medical needs. The Company’s approach consists of focusing on cancer’s metabolism through disruption of protein synthesis and increasing oxidative stress, which it believes provides improvement over conventional treatment solutions, as it targets the underlying cause of the cancer rather than just symptoms. Tyme is investing in new therapeutic approaches such as amino acid transport, disrupting critical protein synthesis, modulating autophagy and inhibiting key intracellular pathways.

Image Source: Company
The Company’s lead product candidate is SM-88, which is being developed for the treatment of multiple advanced cancers such as pancreatic, prostate, sarcoma and breast among others. SM-88, when used in combination with MPS, has the potential to interrupt protein synthesis, reduce key cellular defense and cause oxidative stress-related apoptosis. In addition, the candidate has demonstrated a differentiated mechanism of action (MOA) and is ideal for combining with existing therapies, besides showing monotherapy activity in both solid and hematological cancers. The candidate is currently undergoing a pivotal trial and an adaptive randomized Phase II/III clinical trial in patients with second- and third-line pancreatic cancer. In addition, SM-88 is also being evaluated in a Phase II study for the treatment of high-risk sarcomas. SM-88 has shown clinical response across 15 tumor types, where imaging responses were demonstrated.
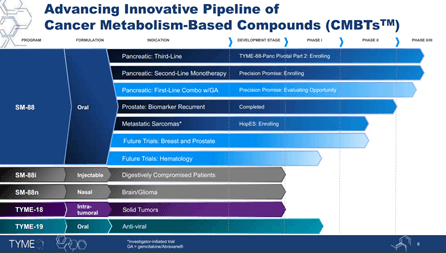
Image Source: Company
Furthermore, the Company is developing TYME-19, a synthetic bile acid, to be used as a metabolic agent for the treatment of COVID-19. The Company is planning to initiate a proof-of-concept trial in partnership with physicians from Massachusetts General Hospital and the Weill Cornell Medical Center and is awaiting approvals for customary trial sites.
The Company has a robust intellectual property rights portfolio with over 200 issued patents and patent applications worldwide that cover compositions, methods, manufacturing which offer protection until 2032.
Tyme has multiple inflection points lined up for 2021, including enrollment in TYME-88-Panc pivotal study and the HopES Phase II Trial, PanCAN’s Precision PromiseSM adaptive randomized Phase II/III Trial in patients with pancreatic cancer. The Company intends to present clinical data from TYME-88-Panc Study at the ASCO GI 2021 Symposium scheduled for H2,2021. Additionally, Tyme is planning the publication of additional data from the clinical trials of SM-88 and Initiate TYME-19 proof of concept trial in the second half of 2021.
Agenus Bio (NASDAQ: AGEN)
Market Cap: $947.60M; Current Share Price: 4.26 USD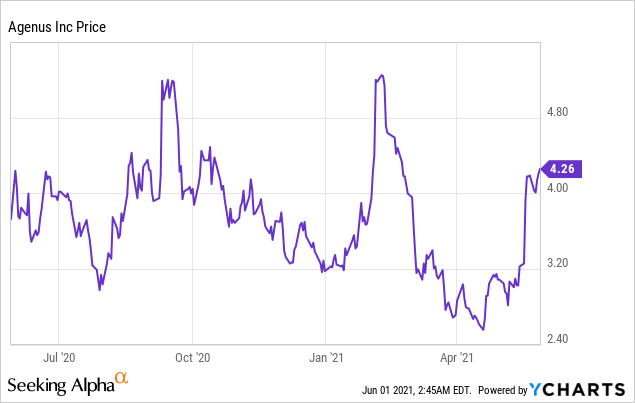
Data by YCharts
Agenus is developing a pipeline of immune-oncology products that comprises checkpoint antibodies, cell therapies, vaccines and adjuvants that aim to combat cancer by delivering innovation and optimal drug combinations. The Company is leveraging its world-class scientific team, unique technology platform, research and GMP manufacturing capabilities to understand patient requirements and create curative treatments. The company is working on both single agent and combination therapies to target multiple mechanisms such as stimulating the immune system through cancer vaccines, mobilizing immune cells by using priming agents, blocking immune resistance using immune checkpoints and developing bispecifics that can condition tumor microenvironment.
The Company’s lead product candidate is Zalifrelimab [AGEN1884] + balstilimab [AGEN2034], an anti-CTLA-4 antibody in combination with anti-PD-1 antibody, intended for the treatment of second-line cervical cancer and AGEN1181, a second generation anti-CTLA-4 antibody) being developed as a monotherapy and in combination with balstilimab.
The Company has recently entered into a definitive agreement with Bristol Myers Squibb for AGEN1777, a proprietary bispecific antibody program, which can block TIGIT and a second undisclosed target. Agenus will receive $200 million upfront payment and up to $1.36 billion in development, regulatory and commercial milestones in addition to tiered double-digit royalties on net product sales as per a Company statement.
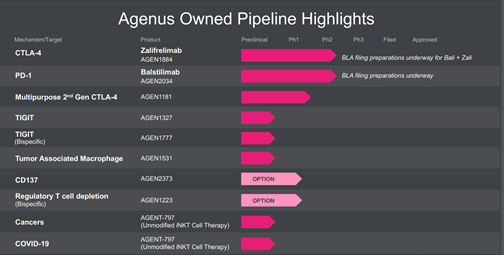
Image Source: Company
Agenus is planning to submit an Investigational New Drug application with the FDA for AGEN1777 in H2,2021 and intends to develop the candidate for the treatment of high priority tumor indications such as non-small cell lung cancer.
Furthermore, the Company is developing AGEN2373, a fully human monoclonal antibody in collaboration with Gilead, besides two more programs namely AGEN1223, a bispecific designed to selectively deplete immunosuppressive T regulatory cells from the tumor microenvironment and AGEN1423, a TME conditioning Anti-CD73/TGFβ TRAP Bifunctional Fusion Proteinβ TRAP Bifunctional Fusion Protein.
Agenus has a global alliance with INCYTE, through which it is developing INCAGN1876, a GITR (glucocorticoid-induced TNFR-related protein) agonist; other programs include a Phase 1 trials of INCAGN1949, INCAGN2390, and INCAGN2385 targeting OX40, TIM3, a checkpoint inhibitor, LAG-3 (lymphocyte-activation gene 3) a checkpoint protein respectively, there is another undisclosed target under evaluation as well.
The Company has developed unparalleled capabilities in research, development and manufacturing and has expertise in areas such as immunology, structural biology and computational biology. Agenus also has an in-house cell line development that is capable of delivering monoclonal, stable and have commercial grade yields for conducting preclinical and commercial supply.

Image Source: Company
Agenus is also working on creating personalized vaccines namely Prophage and AutoSynVax that contain cancer neoantigens, using its proprietary Agenus Immunogenic Mutation (AIM™) algorithms and Next Generation Sequencing (NGS) technologies, cloud computing and our cutting-edge proprietary bioinformatics capabilities.
Agenus’s proprietary QS-21 Stimulon™, a key component of GlaxoSmithKline’s (GSK’s) Shingrix, received $15.1M milestone payment from HealthCare Royalty Partners (HCR) based on net sales of vaccine exceeding $2.0B in 2019. The Company is also eligible for another milestone payment of $25.5M, if Shingrix net sales exceed $2.75B over four consecutive quarters before 2026.
The Company has two state-of-the-art in-house GMP manufacturing facilities located in Berkeley, CA (Agenus West) and Lexington, MA.
Disclosure: I/we have no positions in any stocks mentioned, and no plans to initiate any positions within the next 72 hours.
Click here to please visit our detailed disclosure
References
https://www.pieris.com/pipeline/overview
https://www.syros.com/platform
https://www.xxiicentury.com/technology/our-tobacco-technology
https://www.xxiicentury.com/products/vln
https://s22.q4cdn.com/265040820/files/doc_presentations/2021/TYME_CORP_FEB2021.pdf
https://investor.agenusbio.com/static-files/a9a44240-d6c3-4f69-82da-89bfd927c9f8





No Comments