
23 Aug Should Gritstone Bio be a part of your Investment Portfolio?
Gritstone Bio, Inc. (NASDAQ: GRTS), formerly known as Gritstone Oncology, is a clinical-stage biotechnology company developing targeted immunotherapies for a range of diseases in the cancer and infectious disease space. The Company’s approach is based on leveraging the inherent abilities of the immune system to target and destroy select antigens. While the initial focus of the Company was on tumor-specific antigens, the same has now been extended to include viral antigens.
The Company’s proprietary EDGE platform enables the delivery of patient-specific neoantigens most likely to be present on the tumor. The use of inhouse sequencing on a routine clinical biopsy helps devise a set of personalized predicted neoantigens that can be administered by an intramuscular injection.
The Company was founded in 2015 and is headquartered in Emeryville, California.
Gritstone Bio Inc (NASDAQ: GRTS)
Market Cap: $372.34M; Current Share Price: 7.53 USD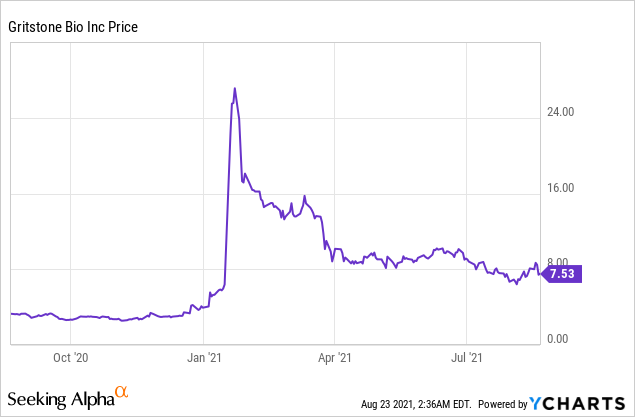
Data by YCharts
Strengths
The Company’s pipeline consists of two main focus areas namely, Oncology and Infectious Diseases. Gritstone is developing a second-generation vaccine targeting SARS-CoV-2, the virus that causes COVID-19, which has the potential to provide longer-lasting protection and potency and most importantly acts against the emerging Spike mutations of SARS-CoV-2.
The Company has entered into a licensing agreement with La Jolla Institute for Immunology, for access to validated SARS-CoV-2 antigens. Gritstone is using its Edge and vaccine platform technologies to develop a novel vaccine containing spike and other viral antigens that provide good targets for T cell immunity. The Company’s vaccine has the potential to protect against future coronavirus pandemics.
The candidate is currently being evaluated in a Phase 1 clinical trial, which is being conducted through the Infectious Diseases Clinical Research Consortium (IDCRC), with support from The National Institute of Allergy and Infectious Diseases (NIAID), part of the National Institutes of Health. The Company has received a grant from the Bill and Melinda Gates Foundation for the preclinical evaluation of the candidate.
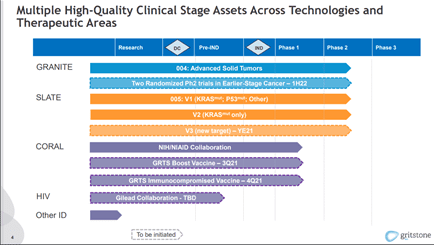
Image Source: Company
The Company has a collaboration with Gilead Sciences for developing a HIV-specific vaccine based-immunotherapy that uses the company’s proprietary prime-boost vaccine platform, for treatment of human immunodeficiency virus (HIV) infection. The Companies are now planning to initiate a Phase 1 clinical trial based on encouraging preclinical results.
The collaboration with Gilead involves an upfront payment of $ 60 million, with a total deal value of up to $785 million plus royalties, based on achievement of certain clinical, regulatory and commercial milestones.
As part of its oncology portfolio, the Company is evaluating GRANITE, a personalized neoantigen-based immunotherapy for the treatment of microsatellite stable colorectal cancer (MSS CRC) who have progressed on FOLFOX/FOLFIRI therapy and a second cohort for patients with gastro-esophageal cancer who have progressed on chemotherapy as per the company. GRANITE has been awarded a fast-track designation by the U.S. Food and Drug Administration for the treatment of MSS CRC.
The candidate is currently undergoing the Phase 2 portion of a Phase 1 / 2 study in combination with checkpoint inhibitors.
The Second product SLATE is a shared neoantigen immunotherapy that uses a fixed set of neoantigens common to certain cancer patients, providing an off-the-shelf alternative. The candidate is being studied in a Phase 2 portion of a Phase 1 / 2 clinical trial in combination with checkpoint inhibitors, targeting the treatment of non-small cell lung cancer with relevant KRAS mutations who have progressed on prior immunotherapy, and for cancer types where a relevant TP53 mutation exists.
Image Source: Company
Furthermore, Gritstone’s pipeline also consists of a BiSpecific Antibody ((BiSAb) therapeutics. The primary target is CT83, a cancer-germline antigen found on certain solid tumors with limited expression on normal tissues. The candidate is currently being evaluated in IND-enabling studies.
The Company is exploring the application of its proprietary EDGE platform to find targets for T-cell receptor (TCR) directed cell therapies. Gritstone has a collaboration with bluebird bio for identifying tumor-specific targets and natural TCRs that can be used in bluebird bio’s existing cell therapy platforms.
As part of its agreement with bluebird bio, Gritstone will provide 10 tumor-specific targets for multiple tumor types, along with TCRs directed to those targets in certain cases.
The Company has a fully insourced 43,000 sq. ft. manufacturing and testing facility in Pleasanton, CA.
Weakness
The Company does not yet have a successful commercial product in the market. Gritstone is a fairly new entrant to the COVID-19 space and may not be able to commercialize the company’s vaccine candidate during the pandemic. However, the Company’s differentiated mechanism of action that involves targeting the spike as well as the other viral antigens, may prove to be a winner in the long-term, especially when dealing with the emerging variants.
Opportunity
Colorectal Cancers start in the Colon or the Rectum and are often used interchangeably as they share many common features. These usually take the shape of an abnormal cell growth or polyps on the inner lining of the colon or rectum, some of which may turn cancerous, especially if they are Adenomatous polyps (adenomas).
Adenocarcinomas constitute over 96% of colorectal cancers, followed by carcinoid tumors, gastrointestinal stromal tumors (GISTs), Lymphomas and Sarcomas.
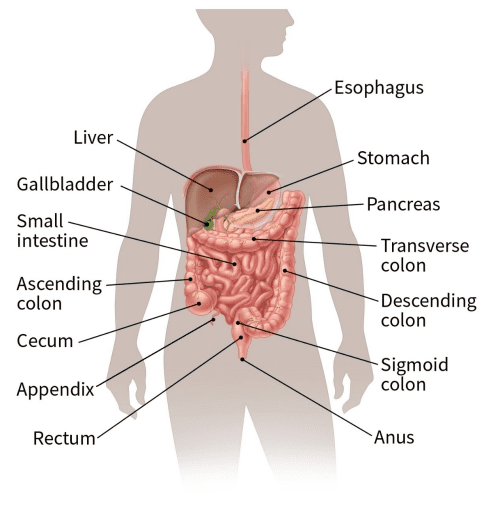
Image Source: Cancer.Org
Colorectal Cancer is the third most common form of cancer diagnosed in the U.S. According to an estimate by the American Cancer Society, about 101,420 new cases of colon cancer and 44,180 cases of rectal cancer will be diagnosed in 2019 in the U.S alone, with over 51,020 patients likely to succumb to it during 2019.
A report by Grand View Research, Inc., predicts that the global colorectal cancer market will grow at a CAGR of 3.3% to reach USD 10.8 billion by 2022. The increasing geriatric population, rise in obesity, diabetes, improper lifestyle habits such as smoking, heavy use of alcohol or a low-fiber, high fat diet are some of the causes for the growing incidences of colorectal cancers. In addition, a personal history of polyps, or family history of this illness increases the risk manifold.
There is growing awareness about this disease coupled with increase in research and development efforts by government and private healthcare entities such as The American Cancer Society, the Centers for Disease Control and Prevention (CDC), and the National Colorectal Cancer Roundtable that intend to have 80% of adults age 50 and older screened for colorectal cancer by 2018. Additionally, The American Cancer Society spent over US $54 million on 92 research grants till March 2018.
Lung cancers are categorized into Non-Small Cell Lung Cancer, the most common form of cancer with an incidence rate of over 85% that manifests as Adenocarcinoma, Large Cell Carcinoma and Squamous Carcinoma. Small cell lung cancer also known as Oat Cell Cancer, which spreads rapidly makes up for around 10% to 15% of the cases and finally Lung carcinoid or neuroendocrine tumors that are less than 5% of the diagnosis and grow slowly and rarely spread.
Lung cancers constitute 13% of all new cancer diagnosis and are the second most common cause of cancer. According to an estimate by the American Cancer Society, there will be over 228,150 new cases of lung cancer (116,440 in men and 111,710 in women) diagnosed in 2019, with about 142,670 deaths resulting from it eventually. The death rate for Lung cancer alone is higher than that of colon, breast, and prostate cancers combined.
Image Source: Freepik
The Global Lung Cancer market is estimated to grow at a CAGR of 13.2%, during 2018-2022, to reach 36.93 billion by 2022 according to a report by Market Research Future. The rise in geriatric population, pollution levels, negative lifestyle conditions such as Active and passive smoking, increased healthcare expenditure and rapid scientific and technological advancements will drive the growth in the market.
Threats
The Coronavirus Vaccine space has seen a number of companies vying with each other in a race to develop the best vaccination for treating COVID-19. However, with the likes of Pfizer, BioNTech, and AstraZeneca pipping others to the post, the focus is now on a vaccine that may be effective against the rapidly emerging new variants of the disease. There is a possibility that what may work now for one variant may prove to be ineffective in treating the future variants.
Clinical Trials are fraught with risk and uncertainty. There is a possibility that the candidates in the Company’s developmental pipeline may not be able to meet their clinical endpoints in trials. However, a diverse pipeline will help mitigate the risk in case of adverse results or the failure to meet endpoints in any of its ongoing trials. The success of its clinical trials will help the Company advance its pipeline but it should also be prepared to face any setbacks, in case its ongoing trials fail to meet their endpoints.
Conclusion
Gritstone plans to study the efficacy of its vaccine candidate as a booster shot, to be used after vaccination with another currently approved vaccine. This step helps the company address two different markets, those needing complete vaccination and those who only need a booster shot.
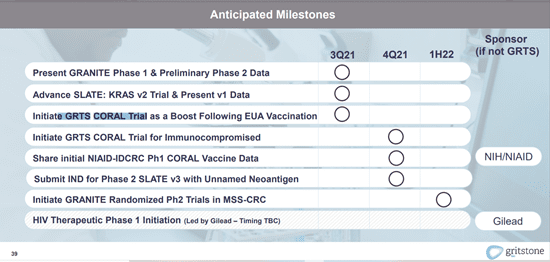
Image Source: Company
Major upcoming catalysts include results from the GRANITE phase 1 and preliminary phase 2 data, expected to be announced in 3Q,2021. In addition, the Company will also be announcing results from the advanced SLATE trial and initiate a GRTS CORAL Trial in the third quarter of 2021.
Furthermore, Gritstone has entered into a funding agreement with CEPI (the Coalition for Epidemic Preparedness Innovations) for the development of a self-amplifying mRNA vaccine candidate. The Company will receive up to US$20.6 million for funding a phase 1 trial.
Disclosure: I/we have no positions in any stocks mentioned, and no plans to initiate any positions within the next 72 hours.
Click here to please visit our detailed disclosure
References
https://gritstonebio.com/our-pipeline/
https://ir.gritstonebio.com/static-files/15857056-7494-41ed-ace0-44026e604210





No Comments