
17 Jan Can Immunome Bounce Back from Clinical Hold on IMM-BCP-01?
Immunome, Inc. (NASDAQ: IMNM), a biopharmaceutical company developing novel first-in-class antibody therapeutics targeting oncology and infectious diseases, announced the receipt of a clinical hold letter from the U.S. Food and Drug Administration (FDA) for its recently submitted Investigational New Drug (IND) for IMM-BCP-01, a three-antibody cocktail, intended for the treatment of SARS-CoV-2 (COVID-19). The FDA has asked for further information related to the preparation and administration of IMM-BCP-01 at clinical sites.
The FDA has also provided guidelines for the clinical protocol for IMM-BCP-01, which include patient selection criteria. The Company is working on providing the requisite information quickly. The Company had submitted an Investigational New Drug (IND) application for IMM-BCP-01 in November 2021.
IMM-BCP-01 is a three-antibody cocktail that works on the non-overlapping regions of the Spike protein of SARS-CoV-2, elicits ACE2 and non-ACE2 dependent neutralization and induces natural viral clearance mechanisms. The candidate has demonstrated the ability to significantly reduce viral load in lungs in animal testing and has the potential to broadly neutralize the Delta (B.1.617.2) variant in vitro, with the Company engaged in testing against the Omicron variant.
Purnanand Sarma, PhD, President & CEO of Immunome, commented,
“We are thankful for the rapid and insightful feedback that the FDA has provided throughout our ongoing discussions and have already initiated efforts to provide the requested information.”
The candidate’s development was funded by the U.S. Department of Defense’s (DOD) Joint Program Executive Office for Chemical, Biological, Radiological and Nuclear Defense (JPEO-CBRND) in collaboration with the Defense Health Agency (DHA).
The Company intends to report data from IMM-BCP-01’s activity against the Omicron variant in live virus testing, conducted by a third-party lab in January and is confident about the candidate’s efficacy.
Immunome, Inc. (NASDAQ: IMNM)
Market Cap: $129.11M; Current Share Price: 10.67 USD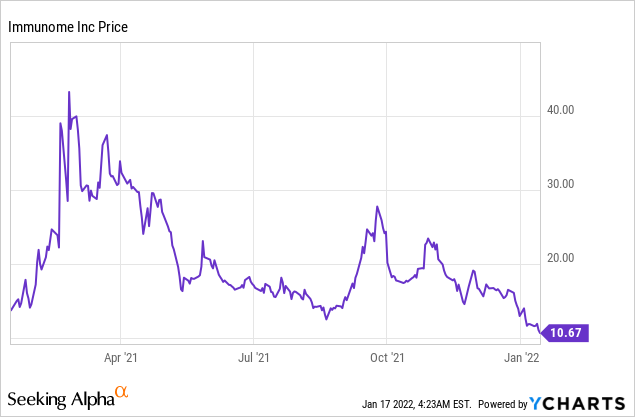
Data by YCharts
Opportunity
According to the World Health Organization (WHO), Coronavirus (CoV) is a family of viruses that cause illnesses ranging from common cold to lethal diseases such as the Middle East Respiratory Syndrome (MERS-CoV), Severe Acute Respiratory Syndrome (SARS-CoV) and a new strain of CoV named, novel coronavirus (nCoV). The virus is Zoonotic as it is transmitted from animals to humans, with the presence of many strains that have not yet infected humans. The infection manifests in the form of respiratory symptoms, cough, fever, shortness of breath and can take the form of pneumonia, severe acute respiratory syndrome, kidney failure and even death.
There is no definite cure or treatment for eradication of CoV, with treatment being limited to alleviation of symptoms, and providing support and care to patients. Scientists have been able to sequence the virus genetic code; spurring organizations around the world into action.
The COVID-19 pandemic is likely to become endemic, similar to the seasonal FLU and countries have already undertaken vaccinations on a war-footing, even in less developed countries. The initial race to create vaccinations is now over, with some emerging as clear winners, while others ruin a missed opportunity.
However, new variants are emerging, the most recent being Omicron, which was designated as a variant of concern by the WHO on 26 November 2021. Omicron has several mutations and is currently spreading rapidly across the world, reinfecting those previously affected by COVID-19 as well. In view of the same, WHO has recommended countries to enhance surveillance, undertake sequencing of cases and share them on public databases such as GISAID, besides performing field and laboratory assessments.
Company
Immunome leverages its Discovery Engine to extract information present in “highly-educated” B cells, in patients who have the ability to fight their illnesses. The approach enables the discovery of antigens and native-antibodies obtained from patients, which can bind to these targets with high affinity.
The Company’s approach is an improvement over conventional one’s as it enables the identification of antibodies with the highest potential to combat cancer and infectious diseases. Immunome has been able to interrogate more than 150 patient samples and produce more than 250,000 hybridomas from patients.
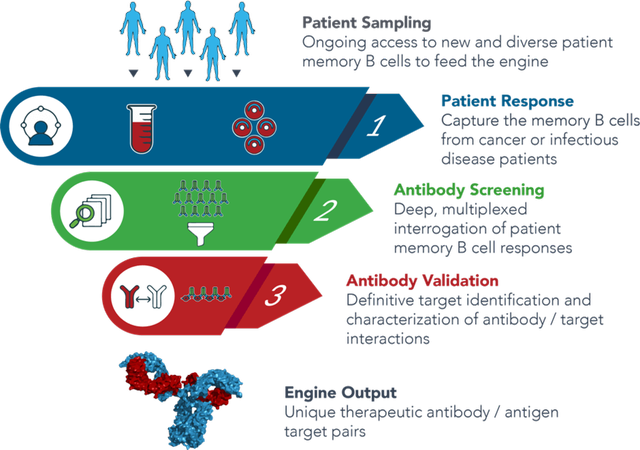
Image Source: Company
The technology is capable of screening up to 20,000 antibodies in a single experiment and identifying novel antigen targets and antibodies that bind with high affinity. The Company is actively seeking partnerships with pharmaceutical companies, government agencies and global health organizations to develop novel therapeutics with significant impact on human health.
The Company’s pipeline consists of two candidates namely IMM-BCP-01 for infectious diseases such as Three SARS CoV-2 Epitopes and IMM-ONC-01, an oncology candidate targeting IL-38.
The Company intends to file an Investigational New Drug application (IND) for IMM-ONC-01 in the first quarter of 2022 and is currently evaluating the candidate in preclinical studies. The candidate is a member of the IL-1 family of cytokines and has the potential to block the IL-38 function, which is responsible for tumor cells evading the immune system response efforts.
The Company’s platform has produced multiple non-canonical targets that reveal a “functional clustering” of targets that are a subject of immune response by patients, providing valuable insights into the tumor-driven processes to be targeted to tackle diseases.
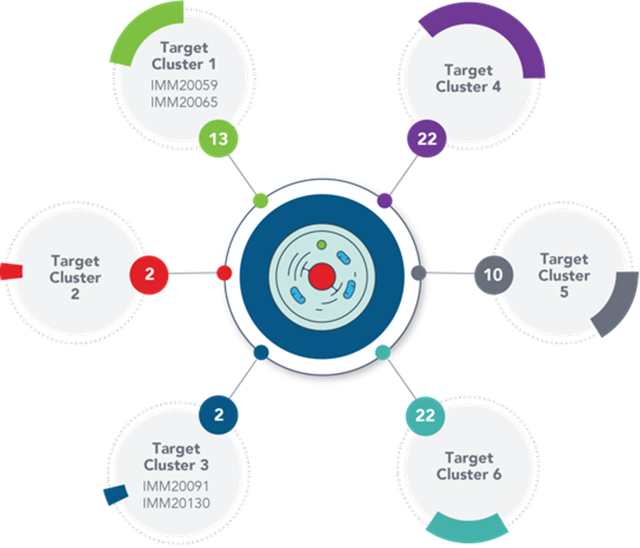
Image Source: Company
Immunome is currently engaged in creating a mixture of antibodies for a Biosynthetic Convalescent Plasma (BCP), intended to be used in both treatment and prophylactic settings.
Key Takeaways
Disclosure: I/we have no positions in any stocks mentioned, and no plans to initiate any positions within the next 72 hours.
Click here to please visit our detailed disclosure
Reference
https://www.who.int/news/item/28-11-2021-update-on-omicron
https://www.immunome.com/about-us/
https://www.immunome.com/pipeline/





No Comments