
13 Mar Amphastar: New FDA Approval Opens a Floodgate of Opportunities
Amphastar Pharmaceuticals, Inc. (NASDAQ: AMPH) recently announced that the U.S. Food and Drug Administration has granted approval of its New Drug Application (NDA) for naloxone hydrochloride nasal spray 4mg, which is delivered utilizing the Company’s proprietary nasal delivery device. Naloxone hydrochloride nasal spray 4mg is indicated for the emergency treatment of known or suspected opioid overdose, as manifested by respiratory and/or central nervous system depression for adult and pediatric patients.
Dr. Jack Zhang, Amphastar’s President and Chief Executive Officer commented:
“The approval of our naloxone hydrochloride nasal spray marks an important step in helping alleviate the opioids crisis as patients can further broaden their access to a critical product.”
According to IQVIA, the U.S. sales of naloxone hydrochloride nasal spray 4mg in 2022 was approximately $250 million for the 12 months ended December 31, 2022.
The approval of naloxone hydrochloride nasal spray by the FDA is quite significant for Amphastar as it not only widens their product portfolio and revenue opportunities, but it also validates their technical capabilities, and allows the Company to leverage it towards their intranasal pipeline.
Amphastar Pharmaceuticals (NASDAQ: AMPH)
Market Cap: $1.71B; Current Share Price: $35.73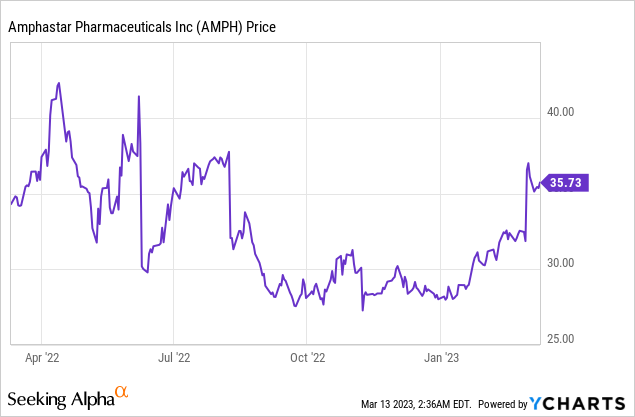
Data by YCharts
The Company
Amphastar Pharmaceuticals is a bio-pharmaceutical company that focuses primarily on developing, manufacturing, marketing, and selling technically challenging generic and proprietary injectable, inhalation, and intranasal products, and insulin active pharmaceutical ingredient, or insulin API products.
Amphastar is currently developing a portfolio of generic abbreviated new drug applications, or ANDAs, biosimilar insulin product candidates and proprietary product candidates, which are in various stages of development and target a variety of indications.
Product Portfolio
Amphastar manufactures and sells over 20 products, the overwhelming majority of which are prescription pharmaceuticals. Their product portfolio includes injectable and metered-dose inhalers (MDI) products; along with complex, combination products.
Some of the indications addressed by Amphastar’s products include deep vein thrombosis, asthma, opioid overdose, pain management, anesthesia, and hypoglycaemia. These are managed by drugs such as Enoxaparin, Glucagon, Lidocaine Injection and Jelly, Vitamin K1, Naloxone Commercial Product Portfolio, Primatene MIST®, Epinephrine PFS & MDV, Cortrosyn®, Ganirelix, Vasopressin.
The Company’s largest products by net revenues currently include Primatene MIST®, epinephrine, glucagon, phytonadione, lidocaine, and enoxaparin sodium. Overall, revenues from drug sales are well diversified over a strong and wide range of existing products.
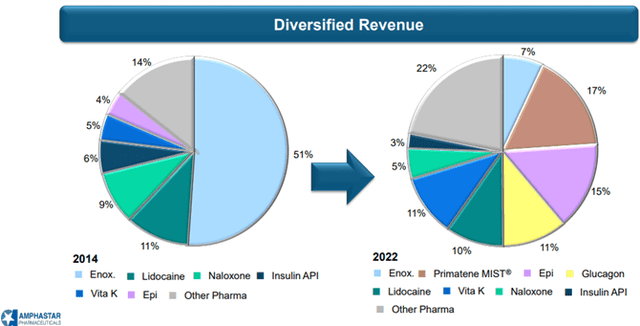
Image Source: Company
We’ll discuss 4 key elements which show that Amphastar seems to have a bright future:
- Operates in Markets with High Technical Barriers to Entry
Amphastar aims to serve large markets with high technical barriers to entry. The Company is specifically focused on products that leverage their proprietary research and development capabilities, require raw materials or APIs for which the Company has a competitive advantage in sourcing, synthesizing, or manufacturing; and/or improve upon an existing drug’s formulation with respect to drug delivery, safety, and/or efficacy.
Amphastar already has multiple technological capabilities that enable the development of technically challenging products with limited competition. These capabilities include characterizing complex molecules, analyzing and synthesizing peptides and proteins, conducting immunogenicity studies, engineering particles, and improving drug delivery through sustained-release technology.
Through these technological capabilities the Company produces bioequivalent versions of complex drugs and supports the development and manufacture of a broad range of dosage formulations, including solutions, emulsions, suspensions, and lyophilized products, as well as products administered via pre-filled syringes, vials, nasal sprays, metered-dose inhalers, or MDIs, and dry powder inhalers, or DPIs.
Amphastar is especially focused on the injectable and inhalation markets.
Based on a December 2022 IQVIA National Sales Perspective Report, the U.S. injectable drug market in 2022 was over $285 billion, while the inhalation drug market was approximately $29 billion. Amphastar’s generic development portfolio is targeting opportunities in over $16 billion and $6 billion of these markets respectively.
Both the injectable and inhalation product markets have high barriers to entry – there are a limited number of companies with the technology and experience needed to manufacture these products, hence Amphastar is well poised to benefit from the above opportunities.
- Strong Focus on R&D Investment
Amphastar has a fully covered R&D team from early stage to clinical trial and from laboratory to scale-up – as of December 31, 2022, the Company had 249 employees dedicated to research and development with expertise in areas such as pharmaceutical formulation, process development, toxicity studies, analytical, synthetic, and physical chemistry, drug delivery, device development, equipment and engineering, clinical research statistical analysis, etc.
Additionally, in the recent past, the Company invested heavily in research and development – for the years ended December 31, 2022, 2021, and 2020, Amphastar’s research and development expense was $74.8 million, $60.9 million, and $67.2 million, respectively.
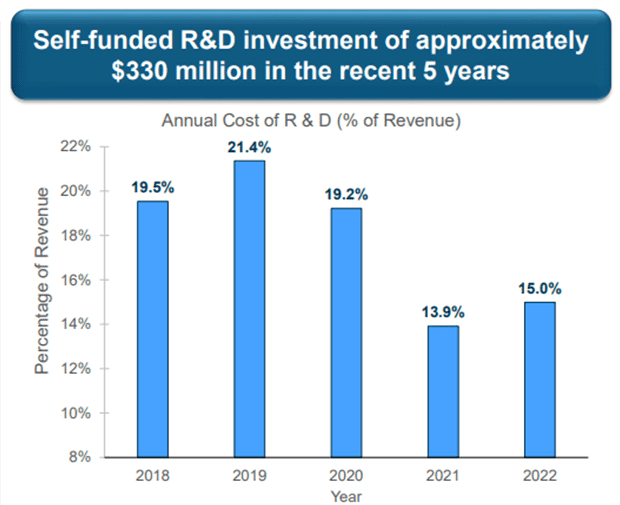
Image Source: Company
The Company expects to continue to make substantial investments in research and development to expand product portfolio and grow the business. Their aim is to ensure diverse pipeline development with flexibility and scalability for sourcing API, starting material, and research under their vertically – integrated platform.
Amphastar’s emphasis on research and development will enable it to improve its product portfolio and keep it one step ahead of the competition, thereby assisting in the long-term growth of the Company.
- Robust Revenue Growth
For FY22, the Company reported net revenues of $499.0 million, and GAAP net income of $91.4 million, or $1.74 per share, compared to net revenues of $438 million and GAAP net income of $63.3 million, or $1.25 per share for FY22.
The increase in revenue was primarily due to increase in unit volumes for dextrose and sodium bicarbonate, which were in high demand due to competitor shortages, higher unit volumes of isoproterenol, and launches of ganirelix and vasopressin, in June 2022 and August 2022, respectively.
Products such as Primatene MIST®, Epinephrine, and Glucagon also showed increase in unit volumes – all of which indicate Amphastar’s product popularity and growing market share.
In fact, over the last five years, the Company’s sales and gross profit have shown consistent upward growth, increasing by as much as 14%/ year and 23%/ year respectively.
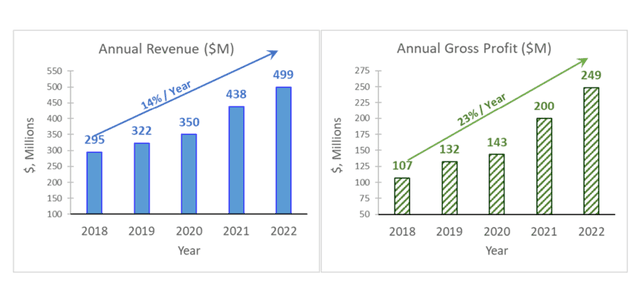
Image Source: Company
Given the Company’s diversified and strong base of existing products, along with the promising products that Amphastar has in its pipeline, it seems that its revenues are set to only increase further.
- Encouraging Product Pipeline and Growth Drivers
The Company currently has three ANDAs on file with the FDA targeting products with a market size of over $3 billion, three biosimilar products in development targeting products with a market size of over $11 billion, and six generic products in development targeting products with a market size of over $9 billion. This market information is based on IQVIA data for the 12 months ended December 31, 2022. The Company is developing multiple proprietary products with injectable and intranasal dosage forms.
Over time, Amphastar’s pipeline projected to advance with more focus on proprietary and biosimilar products.
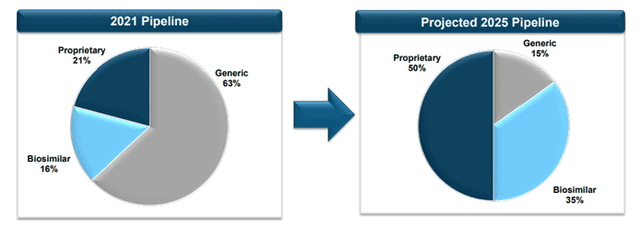
Image Source: Company
At the moment, in the generic pipeline, the Company has 10 candidates with technical barriers.
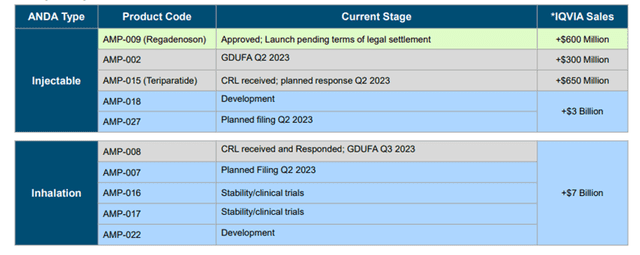
Image Source: Company
Their diabetes portfolio consists of the first and only FDA approved generic Glucagon, that covers the full spectrum of the insulin from rapid to long acting.

Image Source: Company
The AMP-004 BLA planned filing is in 2023, and indicates $12 Billion in IQVIA sales as of December 2022, ~125 million units of both pens and vials.
Other key milestones for 2023 include
- AMP-027 planned filing Q2 2023
- AMP-007 planned filing Q2 2023
- AMP-015 (Teriparatide) CRL received; planned response Q2 2023,
And expected approvals of
- Intranasal Naloxone refiled September 2022; PDUFA Q1 2023
- AMP-002 CRL received; GDUFA Q2 2023
- AMP-008 CRL received; GDUFA Q3 2023
For 2023, the key growth drivers are expected to include the glucagon injection kit, which has exhibited strong sales since launched in Feb. 2021, especially given the current increased market opportunity and the Company’s approved increased capacity.
The Drug Vasopressin, launched in August 2022, is also expected to contribute to increased revenues, along with Primatene MIST®, which witnessed a price increase in January 2023, and whose Nationwide TV, Radio and Digital Advertising are driving weekly sales.
To summarize, Amphastar seems to be on an upward trajectory due to its continuously increasing revenues, promising product pipeline and robust research and development strategy – as already mentioned, the FDA recently granted approval for its New Drug Application and there are more to follow. Additionally, Amphastar faces little competition as it operates in a high barrier to entry industry – and all of these factors indicate that the Company is worth watching out for.
Disclosure: I/we have no positions in any stocks mentioned, and no plans to initiate any positions within the next 72 hours.
Click here to please visit our detailed disclosure
Reference:
https://www.sec.gov/ix?doc=/Archives/edgar/data/1297184/000129718422000056/amph-20220930x10q.htm
https://www.sec.gov/ix?doc=/Archives/edgar/data/1297184/000129718423000019/amph-20221231x10k.htm





No Comments