
06 Dec Artivion: Growing Aggressively in Cardiac Health Market
Artivion, Inc. (NYSE: AORT) is a leader in the manufacturing, processing, and distributing medical devices and implantable human tissues used in cardiac and vascular surgical procedures for patients with aortic disease. The Company aims to chart a path from profitable stability to aggressive, predictable growth.
Artivion, Inc. (NYSE: AORT)
Market Cap: $742.98M; Current Share Price: 17.73 USD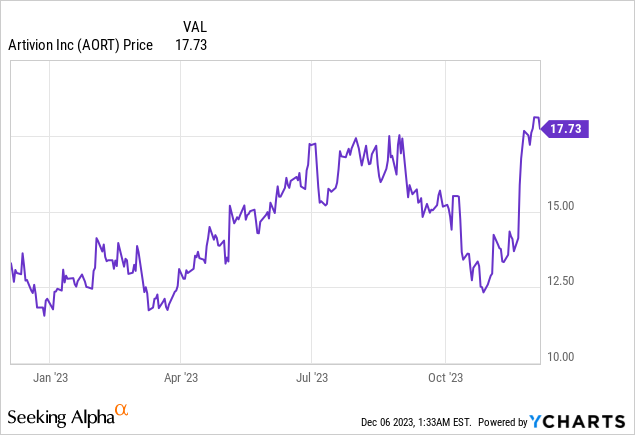
Data by YCharts
The Company and its Products
Artivion has four primary product families: aortic stent grafts, surgical sealants, On-X® mechanical heart valves, related surgical products, and implantable cardiac and vascular human tissues. Aortic stent grafts include aortic arch stent-grafts, abdominal stent-grafts, and synthetic vascular grafts. Aortic arch stent grafts include E-vita Open NEO, E-vita Open Plus, the Ascyrus Medical Dissection Stent (AMDS) hybrid prosthesis, the NEXUS® endovascular stent graft system (NEXUS), and E-vita Thoracic 3G products.
Abdominal stent grafts include E-xtra Design Engineering, E-nside, E-tegra, E-ventus BX, and E-liac products. Surgical sealants include BioGlue® Surgical Adhesive (BioGlue) products. In addition to these four primary product families, the Company sells or distributes PhotoFix® bovine surgical patches, CardioGenesis® cardiac laser therapy, and PerClot® hemostatic powder (before the sale to a subsidiary of Baxter International, Inc.
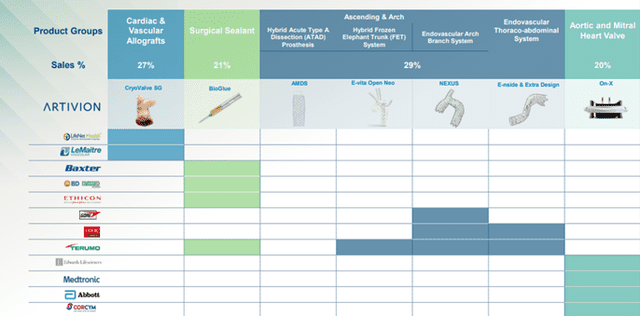
Image Source: Company
Artivion’s principal operating subsidiaries include JOTEC GmbH (JOTEC), a Hechingen, Germany-based endovascular and surgical products company acquired on December 1, 2017, and On-X Life Technologies, Inc. (On-X), an Austin, Texas-based mechanical heart valve company acquired on January 20, 2016, as well as separate country entities to support direct sales operations in Brazil, Canada, Cyprus, France, Greece, Italy, Poland, Spain, Switzerland, and the UK. Additionally, Artivion has entities in Australia, China, Colombia, Hong Kong, Korea, Malaysia, New Zealand, Singapore, Thailand, and Vietnam to provide sales and marketing support for the Asia Pacific (APAC) and Latin America (LATAM) regions.
The Company has two reportable segments organized according to products and services: Medical Devices and Preservation Services. The Medical Devices segment includes revenues from sales of aortic stent grafts, surgical sealants, On-X, and other product revenues. The Preservation Services segment includes services revenues from preserving cardiac and vascular implantable human tissues.
We’ll discuss the critical rationale for covering this Company.
- Significant Market Opportunity
The Company’s medical devices and preservation services are primarily used by cardiac and vascular surgeons to treat patients with aortic disease, including heart valve disease, aortic aneurysms and dissections, and, to a lesser extent, other conditions in cardiac and vascular surgery.
Today, there is significant demand for Artivion’s products, which provides it with a total addressable market (TAM) of more than $5.3 billion.

Image Source: Company
In 2015, the Company operated mainly in the tissue and glue segments with a TAM of $600 million. Over the years, it has expanded its product suite and is scaling globally to achieve an expected CAGR of more than 15% post-2025.

Image Source: Company
In the short term, by FY24, the Company expects to achieve revenues of $400 million, which translates into a CAGR of 10.2%.
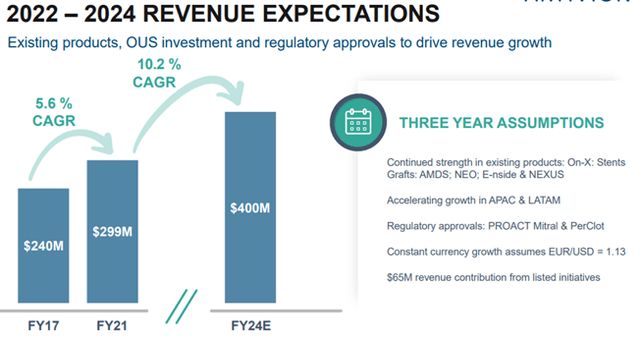
Image Source: Company
The Company has several growth drivers that could help it grab a significant share of the TAM. These include
The above details indicate that Artivion is fully prepared and capable of deriving maximum benefit from the TAM available to it – this, in turn, should help the Company grow by leaps and bounds.
- Robust Strategy
Artivion has a comprehensive strategic plan focused on four growth areas expected to drive the business in the future. These include
- New Products – Through product development and commercialization of new and next-generation products and services focused on aortic repair. As shown below, the Company has a strong product pipeline that provides a $1 billion market opportunity.
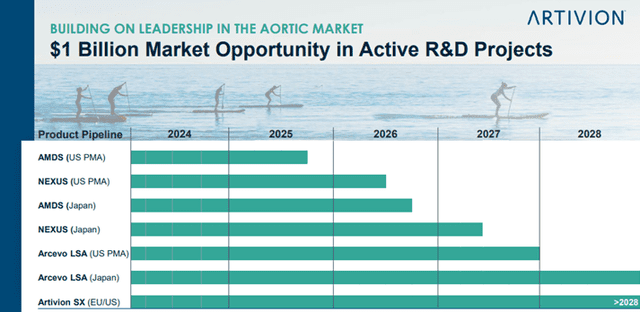
Image Source: Company
- Indications – Through regulatory approvals in new markets and for new products, and through approvals for expanded indications for existing products and services. For example, Artivion has created the first and only FDA-approved mechanical aortic valve for reduced INR.
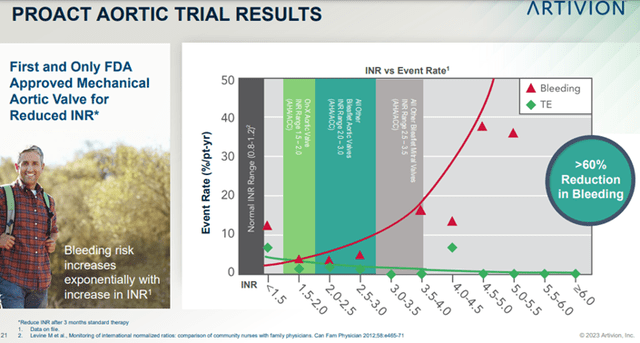
Image Source: Company
- Global Expansion – By entering new international markets, establishing new international direct sales territories, and developing commercial infrastructure in new markets, including emerging markets, such as China and Brazil;
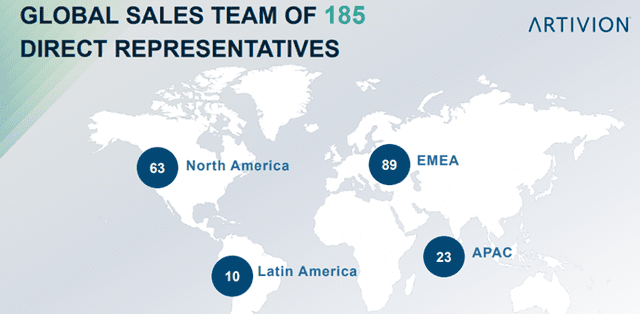
Image Source: Company
- Business Development – By pursuing select acquisitions, licensing, and distribution opportunities aligned to the Company’s objectives and complementing its existing products, services, and infrastructure. Examples include acquisitions of JOTEC, On-X, and Ascyrus Medical LLC (Ascyrus), distribution agreements, and purchase options for NEXUS.
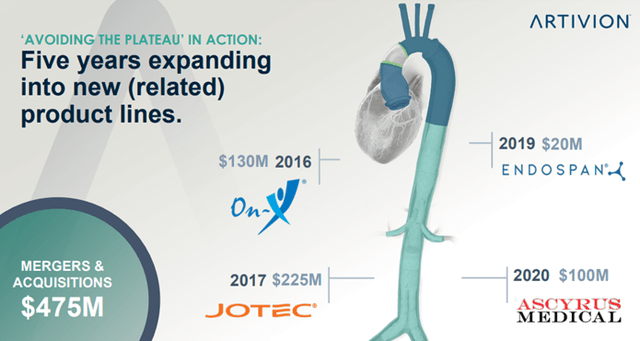
Image Source: Company
Thus, Artivion has an effective strategic plan and is taking adequate steps to implement it to break through to new heights.
- Financial Performance
For Q3 FY23, Artivion achieved revenue of $87.9 million compared to $76.8 million in Q3 FY22, an increase of 14% on a GAAP basis and 12% on a non-GAAP constant currency basis. On-X revenues increased 14% on a GAAP basis and 13% on a non-GAAP constant currency basis in Q3 FY23 compared to Q3 FY22.

Image Source: Company
Non-GAAP adjusted EBITDA increased 34% to $13.9 million in Q3 FY23 compared to Q3 FY22.
Additionally,
Stent grafts: 22% y/y cc revenue growth fueled by robust demand and improved supply • On-X: Low INR label led to 13% y/y cc revenue growth • Preservation services: 12% y/y cc revenue growth driven by pricing initiatives • APAC and LATAM both deliver 20+% y/y cc revenue growth.
For FY22, revenues were $313.8 million, reflecting an increase of 5% on a GAAP basis and 9% on a non-GAAP constant currency basis compared to FY21.
For FY23, Artivion expects its constant currency revenue growth to be between 8.0% and 12.0% compared to FY22 and expects revenues to be between $331 million and $343 million. Artivion expects adjusted EBITDA, as reported, to increase by more than 20% in FY23 compared to FY22, resulting in adjusted EBITDA of more than $50.0 million in FY23.
Thus, the Company is steadily increasing revenues across product segments and geographies. For FY23, Artivion’s revenues and EBITDA are expected to grow steadily, indicating the Company’s continued growth.
Risks
As discussed, Artivion’s robust business strategy brings excellent results and indicates a bright future for the Company. Nevertheless, the Company is exposed to certain risks as well.
Firstly, Artivion’s operations and performance have been and may continue to be impacted by regional and global geopolitical conditions, domestic and foreign trade and monetary policies, and other factors.
Secondly, Artivion operates in highly competitive market segments and faces competition from large, well-established medical device companies and tissue service providers with more significant resources; thus, it may not be able to compete effectively.
Finally, continued fluctuation of foreign currencies relative to the US dollar could materially and adversely affect the business.
Conclusion
Artivion has a global presence with products in high demand, a great business strategy, and steadily increasing revenues, indicating a promising future for the Company. However, it is subject to various risks, such as exposure to regional and geopolitical conditions and highly competitive markets, which could impact its future results. Hence, investors must proceed with caution.
Disclosure: I/we have no positions in any stocks mentioned, and no plans to initiate any positions within the next 72 hours.
Click here to please visit our detailed disclosure
Reference:
https://investors.artivion.com/static-files/002b77b6-2faa-4594-b725-07fb6c7679f8
https://www.sec.gov/ix?doc=/Archives/edgar/data/784199/000078419923000040/aort-20221231.htm
https://www.sec.gov/ix?doc=/Archives/edgar/data/784199/000078419923000150/aort-20230930.htm





Sorry, the comment form is closed at this time.