
16 Mar Can AMX0035 Be a Potential Game Changer in ALS?
Amylyx Pharmaceuticals, Inc. (NASDAQ: AMLX) is a clinical-stage biotechnology Company is focussed on creating novel therapeutics for the treatment of neurodegenerative diseases such as ALS and Alzheimer’s. The Company is awaiting the decision on its NDA for AMX0035 (sodium phenylbutyrate (PB) and taurursodiol (TURSO; also known as ursodoxicoltaurine)) for the treatment of amyotrophic lateral sclerosis (ALS).
Amylyx Pharmaceuticals, Inc. (NASDAQ: AMLX)
Market Cap: $1.37B; Current Share Price: 23.24 USD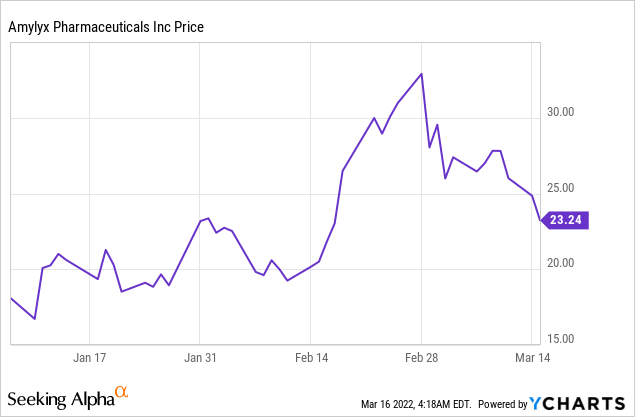
Data by YCharts
We take a holistic look at the Company through a SWOT analysis below:
Company
In December 2021, the Company’s New Drug Application (NDA) for AMX0035 (sodium phenylbutyrate (PB) and taurursodiol (TURSO; also known as ursodoxicoltaurine)) for the treatment of amyotrophic lateral sclerosis (ALS), was accepted by the U.S. Food and Drug Administration. The FDA has granted priority review for the candidate and assigned a Prescription Drug User Fee Act date for AMX0035 of June 29, 2022.
The FDA is going to hold a virtual meeting of the Peripheral and Central Nervous System Drugs Advisory Committee to review the application of AMX0035 on March 30, 2022.
Lahar Mehta, M.D., Head of Global Clinical Development of Amylyx, commented
“There are few treatments approved for ALS, a devastating disease that impacts a person’s ability to move, speak, eat and breathe. We look forward to a robust scientific discussion with the members of the advisory committee panel regarding the clinical data submitted to support our New Drug Application for AMX0035.”
AMX0035, the Company’s lead drug candidate is a combination of sodium phenylbutyrate (PB), a small molecule chaperon that has the potential to lower the unfolded protein response (UPR), thereby preventing cell death and taurursodiol (TURSO; also known as ursodoxicoltaurine), a bax inhibitor that can reduce cell death through apoptosis.
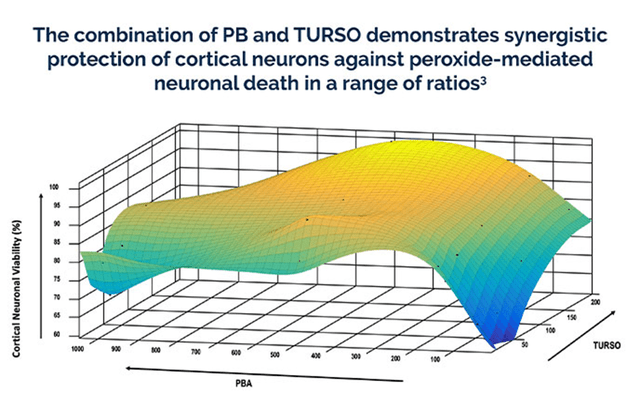
Image Source: Company
The Combination has shown to reduce neuronal death and dysfunction by targeting the key pathways in ALS and other neurodegenerative diseases, namely endoplasmic reticulum and mitochondrial-dependent neuronal degeneration pathways.
In February 2022, the European Medicines Agency’s (EMA) Committee for Medicinal Products for Human Use (CHMP) accepted the Company’s Marketing Authorization Application (MAA) for AMX0035, which is now under CHMP review. The submission is supported by data from a Phase 2 clinical trial named CENTAUR, which evaluated 137 adults with ALS at 25 centers of the Northeast ALS Consortium (NEALS) and demonstrated significant reduction in clinical decline as measured by the Revised ALS Functional Rating Scale (ALSFRS-R). In addition, the survival analysis for participants who were followed for up to 3 years demonstrated 44% lower risk of death compared to those who started on placebo.
The trial was partly funded by a grant from the ALS ACT grant and the ALS Ice bucket Challenge.
Furthermore, the Company is also engaged in a Phase 3 PHOENIX clinical trial, a 48-week global clinical trial evaluating AMX0035 (PB/TURSO) for the treatment of ALS., whose primary efficacy outcome will be a measure of survival and Amyotrophic Lateral Sclerosis Functional Rating Scale-Revised (ALSFRS-R) total score progression. Secondary endpoints include change in slow vital capacity (SVC) and ventilation-free survival rates and patient reported quality of life among other measures.

Image Source: Company
The Company is also evaluating AMX0035 for treatment of Alzheimer’s Disease, which is currently undergoing evaluation by scientific advisors on the basis of results from a Phase 2 PEGASUS trial. The Company reported Phase 2 data from the trial in November 2021.
Axcelis is also pursuing the development of the candidate in Wolfram Syndrome and intends to file an IND in the first half of 2022.
Weakness
The decision to conduct an advisory committee meeting comes, roughly five months later after FDA had asked the Company to conduct an additional trial, to test whether the early data help up in consequent studies. However, the FDA retracted on the earlier demand and stated that a new study won’t be necessary for review of the candidate that has been granted a PDUFA data of June 29, 2022.
Drug development efforts in the field of ALS have been challenging for companies, as the disease progresses rapidly after diagnosis and results in death within 2 years. In October 2021, Biogen’s antisense oligonucleotide therapy, Torgersen, which inhibits mutant superoxide dismutase 1 (SOD1) protein, failed to meet the primary efficacy endpoint in the Phase III VALOR Study. Similarly, in May 2021, Arimoclomol, a candidate being developed by Orphazyme failed in a Phase 3 trial.
The early data from the Company, though not definite, have been deemed significant enough to warrant consideration for an approval in Canada and Europe. However, will the FDA and its advisory committee adopt a lenient view of the data, remains to be seen.
Industry
Amyotrophic Lateral Sclerosis (ALS) or Lou Gehrig’s disease is a progressive neurodegenerative disease, characterized by the atrophy of nerve cells in brain and spinal cord, leading to hardening and scarring in the affected region. As the disease progresses, patients gradually lose the functioning of muscles, including those that control speech, movement and breathing. The disease manifests in two forms, sporadic and familial, with 90 to 95 percent of all cases in the U.S being Sporadic. Familial or the inherited form of the disease constitutes the other 5 to 10 percent.
According to information made available by the ALS association, there are over 16,500 Americans afflicted by this illness and nearly 5000 new cases are diagnosed each year. The life expectancy after diagnosis is merely 2 to 5 years, with an estimated $250,000 being spent on out-of-pocket treatment costs. The Global ALS treatment market is expected to be worth over USD 3.6 billion by 2026, growing at a CAGR of 21.6 percent, from USD 0.75 billion in 2018, according to a report by Data Bridge Market Research.
Threat
The Company’s pipeline primarily consists of only one candidate being evaluated in multiple indications. The other indications in which the candidate is being evaluated are either in pre-IND or Phase 2 trial stage. Clinical Trials are fraught with risk and uncertainty. There is a possibility that the candidates in the Company’s developmental pipeline may not be able to meet their clinical endpoints in trials.
The Company may fail to receive regulatory approval for AMX0035, resulting in a setback for the other candidates in the pipeline as well.
However, a diverse pipeline will help mitigate the risk in case of adverse results or the failure to meet endpoints in any of its ongoing trials. The success of its clinical trials will help the Company advance its pipeline but it should also be prepared to face any setbacks, in case its ongoing trials fail to meet their endpoints.
Key Takeaways
Disclosure: I/we have no positions in any stocks mentioned, and no plans to initiate any positions within the next 72 hours.
Click here to please visit our detailed disclosure
References
https://www.clinicaltrialsarena.com/comment/als-biogen-tofersen-fails-trial/
https://investors.amylyx.com/static-files/8d7188f5-9c9e-4123-8204-e54fab324948





No Comments