
28 May Scholar Rock Holdings has Rock Solid Growth Prospects!
Scholar Rock Holding (NASDAQ: SRRK), a clinical-stage biopharmaceutical Company, announced the grant of a Fast-Track Designation for apitegromab, a selective inhibitor of myostatin activation, intended for the treatment of Spinal Muscular Atrophy (SMA) by the U.S. Food and Drug Administration (FDA). The candidate has previously been granted Orphan Drug and Rare Pediatric Disease designations in the U.S and PRIME and Orphan Medicinal Product designations from the EMA for SMA.
Tony Kingsley, President and CEO of Scholar Rock, commented
“We are delighted to receive Fast Track designation and look forward to working closely with the FDA towards our aim of establishing apitegromab as the potential first muscle-directed therapy for patients with SMA. We believe the recently announced top-line data from the TOPAZ Phase 2 trial showed the transformative potential of apitegromab for patients with SMA.”
The Company is developing Apitegromab for the treatment of Spinal Muscular Atrophy and other Myostatin-related disorders. Positive proof-of-concept data from the interim analysis was released in October 2020, which showed potential therapeutic benefits of inhibiting the activation of latent myostatin with apitegromab. Scholar Rock is currently engaged in an ongoing TOPAZ Phase 2 proof-of-concept trial which is evaluating apitegromab in patients with Type 2 and Type 3 SMA over a 12-month treatment period.
Scholar Rock Holding intends to initiate a phase 3 program in Apitegromab by the end of 2021 and also pursue additional opportunities in other muscular dystrophies such as Becker Muscular Dystrophy and other myostatin-related indications.
Scholar Rock Holding, (NASDAQ: SRRK)
Market Cap: $ 921.64M; Current Share Price: 26.78 USD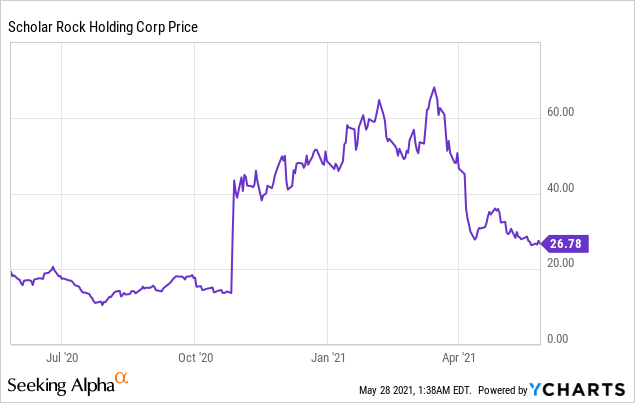
Data by YCharts
Industry
Spinal Muscular Atrophy (SMA), an inherited disorder, is a type of motor neuron disease that causes loss of nerve cells in the spinal cord and brain stem and is characterized by lack of muscle control. The disease is progressive in nature and results in muscle weakness and atrophy in proximal muscles such as back, shoulders and hips. The disease makes it difficult to carry out basic motor functions such as standing, walking, crawling, sitting and in more severe cases even feeding, swallowing and breathing.
The disease can be categorized into Type 1, Type 2, Type 3 and Adult SMA based on the age of onset and severity. SMA is inherited as autosomal recessive genetic disorders and are caused due to the mutations in SMN1 and SMA2 genes. The incidence of the disease is approximately 1 in 10,000 live births and affects one in 8,000 to 10,000 people around the world.
SMA is a degenerative disease and symptoms worsen over time, however medication and physical therapy can improve the condition to an extent. The treatment options for the disease are Spinraza (antisense oligonucleotide) and Zolgensma, a gene therapy.
According to a report by Fortune Business Insights, the global spinal muscular atrophy market is projected to reach USD 14.49 billion in 2026, growing at a CAGR of 28.9 percent, from USD 1.72 billion in 2018. The increase in research and development activities, scientific and technological advancements and improving reimbursement scenario will drive the growth in the market. However prohibitive treatment costs may hamper the progress.
Company
The Company is adopting a unique approach that involves selective targeting of inactive, precursor or latent forms of growth factor, as against the conventional approach of targeting active growth factors. Scholar Rock has applied its novel science across numerous growth factors such as the transforming growth factor beta (TGFβ) and is in pursuit of discovering and developing monoclonal antibodies that can selectively modulate growth factors.
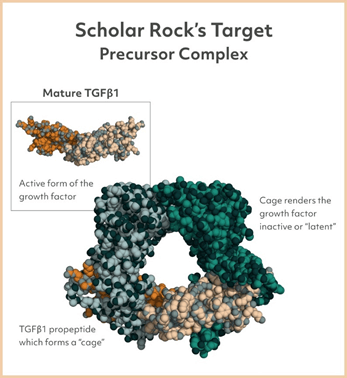
Image Source: Company
Timothy Springer, PhD, co-founder of Scholar Rock has played a crucial role in throwing light on the mechanism of growth factor activation in relation to members of the TGFβ superfamily, by solving a high-resolution X-ray crystal structure of a precursor form of TGFβ1. The TGFβ superfamily has over 30 protein growth factors such as growth differentiation factor (GDF) and bone morphogenetic protein (BMP) that are pivotal to the functioning and regulation of various organs.
Scholar Rock is also developing SRK-181, which has demonstrated that targeting precursor, or latent, form of the TGFβ1 growth factor made tumors vulnerable to anti-PD1 therapy and drive tumor regression through combination therapy and has the potential to make a difference in therapeutics approaches in melanoma, urothelial carcinoma, and breast cancer. The candidate is currently undergoing a phase 1 clinical trial, DRAGON, in patients with locally advanced or metastatic solid tumors.

Image Source: Company
In December 2018, the Company entered into a partnership with Gilead Sciences, Inc (NASDAQ: GILD) to discover and develop selective inhibitors of TGFβ for the treatment of fibrotic diseases. Gilead has worldwide licensing rights to the product candidate that will be developed from three of the TGFβ programs. In January 2020, the Company announced preclinical efficacy in in vivo proof-of-concept studies and is advancing its collaboration with Gilead to determine molecules to be developed as new medicines in fibrotic diseases.
Key Takeaways
Disclosure: I/we have no positions in any stocks mentioned, and no plans to initiate any positions within the next 72 hours.
Click here to please visit our detailed disclosure
References
https://rarediseases.org/rare-diseases/spinal-muscular-atrophy/
https://medlineplus.gov/genetics/condition/spinal-muscular-atrophy/#statistics
https://investors.scholarrock.com/static-files/aac383c1-b3f3-4c0a-ac1e-a1b85b7e556a





No Comments