
13 Apr 5 Biotech Companies That Should Be on Your Investment Watchlist!
Biotech Companies continue to show resurgence even in these challenging times, with more and more companies persevering with bringing their clinical candidates to fruition. While Vaccine development and therapeutics to combat the COVID-19 pandemic are at the forefront of development activities in the biotech sector, there are some companies that are making strides in other related fields such as Oncology, NASH, HBV, and veterinary diagnostics. We bring to you some companies that are leveraging their leading-edge technology to make innovative therapeutics in areas that have a large unmet need.
Arbutus BioPharma Corporation (NASDAQ: ABUS)
Market Cap: $ 303.23M; Current Share Price: 3.11 USD
Data by YCharts
Arbutus Biopharma is a biopharmaceutical company that is engaged in developing a portfolio of drugs with complementary mechanisms that include antiviral action, host targeting and immune based approaches. The Company seeks to revolutionise the HBV treatment landscape and is currently developing therapeutics based on the Nobel-prize winning RNAi mechanism, which uses the body’s own natural processes to eliminate specific gene-products or proteins in the cell.
The Company’s proprietary delivery platform called Lipid Nanoparticle or LNP helps deter the production of disease-associated protein by encapsulating the RNAi trigger molecules in lipid particles and efficiently delivering it into the target cells. Arbutus has a broad pipeline of candidates targeting HBV and has most recently initiated a COVID-19 drug discovery and development program.
Arbutus’s pipeline consists of AB-729, an RNAi that is currently being evaluated in a Phase 1 trial in HBsAg reduction. AB-729 has demonstrated safety, tolerability and a robust and continuous decline in hepatitis B surface antigen (HBsAg) in subjects with chronic hepatitis B (HBV). The Company intends to initiate multiple Phase 2 proof-of-concept combination trials in 2021, and has already started initial screening in a phase 2 proof-of-concept combination trial to evaluate AB-729 in combination with Assembly Biosciences’ lead core (capsid) inhibitor candidate vebicorvir and a nucleos(t)ide reverse transcriptase inhibitor for the treatment of subjects with chronic HBV infection.
AB-836, a capsid inhibitor, is being evaluated in a phase 1 trial for HBV DNA Suppression. The candidate recently received regulatory approval to initiate a phase 1a/1b trial and marks an important step in pursuing future proprietary combinations with AB-729 and other potential mechanisms. In addition, the Company is also developing PD-L1, intended for immune reawakening that is currently in the lead optimization stage.
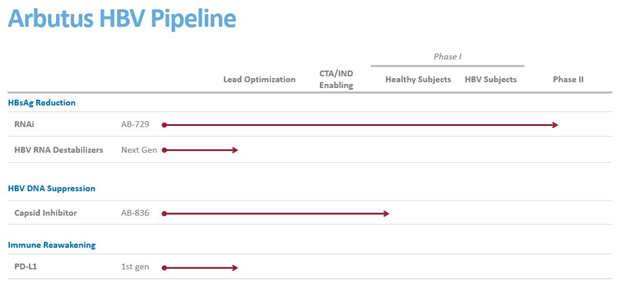
Image Source: Company
In April 2021, the Company entered into a pan-Coronavirus research and license agreement with X-Chem, and Proteros biostructures GmbH (Proteros) for discovery of novel inhibitors targeting the SARS-CoV-2 nsp5 main protease (Mpro). The collaboration will enable Arbutus to screen one of the largest small molecule libraries against Mpro and use state-of-the-art structure guided methods to rapidly optimize Mpro inhibitors, which can then be brought under clinical development.
The Company owns 16% of the common equity of Genevant Sciences Ltd, which was formed in association with Roivant Sciences, Ltd, and has licensed exclusive rights to its lipid nanoparticle and ligand conjugate delivery technologies outside of HBV programs to the company. Arbutus will receive tiered low single-digit royalties on future sales of products that Genevant develops through the licensed patents and can even earn the lesser of twenty percent of the revenue received by Genevant for such sublicensing and tiered low single-digit royalties on product sales by the sublicensee on sub-licensing of its intellectual property.
Arbutus acquired Enantigen Therapeutics, Inc. (Enantigen) in October 2014 for its HBV surface antigen secretion inhibitor program and capsid assembly inhibitor programs. The Company has also entered into an agreement with Gritstone Oncology for use of its lipid nanoparticle (LNP) products and associated intellectual property for development of novel therapeutics for lung and gastric cancer.
In 2018 the FDA and the EMA gave an approval for ONPATTRO™ (patisiran), an RNAi therapeutics intended for treatment of hereditary ATTR amyloidosis with polyneuropathy (FAP) that was developed by Alnylam through licensing rights to Arbutus LNP technology, resulting in a single-digit royalty payments stream to Arbutus. Similarly, the Company is entitled to receive mid-single digit royalty payments for Marqibo, launched by Spectrum Pharmaceuticals (NASDAQ: SPPI), and indicated for the treatment of adult patients with Philadelphia chromosome-negative acute lymphoblastic leukaemia (Ph-ALL)
Hepion Pharmaceuticals, Inc. (NASDAQ: HEPA)
Market Cap: $132.63M; Current Share Price: 1.74 USD
Data by YCharts
A clinical stage biotechnology company, Hepion is developing targeted therapies for liver diseases, such as non-alcoholic steatohepatitis (NASH) and chronic hepatitis virus infection (HBV, HCV, HDV). CRV-431, the Company’s lead drug candidate, is a pleiotropic liver disease drug that binds to cyclophilin isomerase enzymes and inhibits cyclophilin function, far greater than any other cyclophilin inhibitor and has the potential to reduce liver fibrosis and hepatocellular carcinoma tumor. The Company’s therapeutic strategy involves developing drugs that target multiple molecular pathways and have an excellent safety profile with minimal side-effects, besides offering targeted action thereby minimizing the risk of adverse effects.
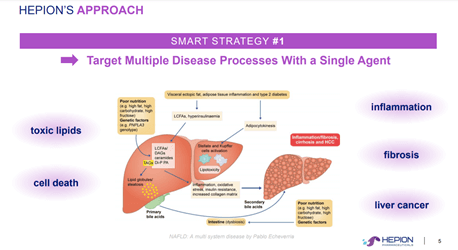
Image Source: Company
AI-Power, Hepion’s proprietary platform is an amalgamation of Artificial Intelligence, Machine Learning and Deep Learning that enables multi-variate, systems-biology bioinformatic and big-data analysis of clinical data with other publicly available databases to improve the drug target selection process, predict drug responders, identify biomarkers and track disease progression and regression. The Company will be leveraging AI Power in a Phase 2a clinical program evaluating CRV431 in non-alcoholic steatohepatitis (NASH).

Image Source: Company
CRV431 has shown an excellent safety profile in animal models and preclinical studies, in addition to having diverse mechanisms of action, 5-times higher accumulation in the liver and antiviral properties. The candidate has also shown to possess antiviral, anti-steatotic, anti-inflammatory, anti-fibrotic, and anti-cancer properties, besides protection from cellular stress and death, which differentiates it from other drug candidates. In March 2021, the Company announced positive results from an in vivo study of CRV431 in a Diet-Induced Animal Model of Non-alcoholic fatty liver Disease namely DIAMOND, that demonstrate CRV431’s potent antifibrotic ability.
The Company is currently engaged in Phase 2a trial to assess the safety, tolerability and pharmacokinetics of CRV431, as standalone and in combination with Tenofovir Disoproxil Fumarate in patients with HBV. Hepion is looking at completing this trial by Q2,20201 and initiating a Phase 2b in Q3,2021.
Hepion’s other areas of therapeutic focus include Hepatocellular Carcinoma and Viral Hepatitis. The core team of the Company has over 30 years of experience in cyclophilin inhibitor development and has discovered and developed the FDA-approved voclosporin for transplantation and autoimmune disease. The Company has a robust intellectual property rights portfolio spanning US, Europe, Australia, Canada, China, Japan and Korea.
Zomedica Corp (NYSE: ZOM)
Market Cap: $1.09B; Current Share Price: 1.12 USD
Data by YCharts
Zomedica is a veterinary diagnostics and pharmaceutical Company creating point-of-care diagnostics products for dogs and cats. In March 2021, the Company sold the very first TRUFORMA® to Guardian Veterinary Specialists, a critical care and specialty hospital in New York. The TRUFORMA allows on-site testing of TSH, total T4, and cortisol assays currently and will include Endogenous ACTH and free T4 in the coming days. The diagnostic platform uses Bulk Acoustic Wave technology developed by Qorvo, to offer an extremely reliable and precise non-optical and fluorescence-free detection system.

Image Source: Company
The TURFORMA diagnostic platform provides sensitive and specific immunoassays that facilitate faster clinical decisions. The platform offers the first feline-optimized TSH assay that can easily differentiate between hyperthyroidism and euthyroid sick syndrome and eliminates the risk of sample transportation, by means of an on-site canine eACTH assay, aiding in the timely diagnosis of adrenal disease. The Company is using the cutting-edge Bulk Acoustic Wave Technology (BAW), that is deployed in aerospace and telecommunications, to deliver reference lab performance levels.
The Company has entered into an agreement with Miller Veterinary Supply for the distribution of TRUFORMA, which is to commence sales from March 30, 2021. Miller, one of the fastest growing businesses in the veterinary industry, will represent Zomedica in multiple states ranging from Texas to Maine and will be aided by Zomedica’s sales representatives along with Miller’s sales personnel in the field.
Zomedica has built an extensive intellectual property portfolio of over 70 patents issued and pending that offer protection to the TRUFORMA diagnostic platform.
Jaguar Health, Inc. (NASDAQ: JAGX)
Market Cap: $217.46M; Current Share Price: 1.70 USD
Data by YCharts
The Company develops plant-based prescription medicine for gastrointestinal disorders through its wholly-owned subsidiary Napo Pharmaceuticals. Mytesi® (crofelemer), a first-in-class anti‑secretory agent developed by Napo Pharmaceuticals, is an FDA-approved treatment for symptomatic relief of noninfectious diarrhea in adults with HIV/AIDS on antiretroviral therapy. Crofelemer, is a plant-based drug extract, derived from the red bark sap of Croton lechleri tree found in the Amazon rainforest. Napo pharmaceuticals has a sustainable harvesting program in place to ensure a product of highest quality that is also sustainable.
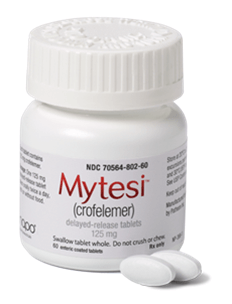
Image Source: Company
The Company holds exclusive worldwide rights for Mytesi and is developing the candidate as potential treatment of follow-on conditions such as short bowel syndrome, inflammatory bowel disease, diarrhea related to cancer therapy, infants with congenital diarrhea issues and idiopathic/functional diarrhea. Jaguar Health is also developing a second-generation proprietary anti‑secretory agent for treatment of diarrhea in Cholera. In addition, the Company is developing a crofelemer delayed-release tablet named Canalevia™ for the treatment of chemotherapy-induced diarrhea (CID) in dogs. The candidate has received a Minor Use in a Major Species (MUMS) designation for CID in dogs and is also aiming at an FDA approval for exercise-induced diarrhea (EID) in dogs.
In September 2020, the Company launched the Entheogen Therapeutics Initiative (ETI) to aid the discovery and development of plant-based medicines derived from psychoactive plants that can act as potential treatment of mood disorders, neuro-degenerative diseases, addiction, and other mental health disorders. Jaguar will be leveraging its proprietary library of 2,300 plants to identify potential development candidates and is focused on finding an alternative to selective serotonin reuptake inhibitor (SSRI) drugs.
Tonix Pharmaceuticals Holding Corp. (NASDAQ: TNXP)
Market Cap: $353.07M; Current Share Price: 1.09 USD
Data by YCharts
The Company is developing a pipeline of candidates focused on small molecules and biologics that can treat central nervous system (CNS) and immunology disorders. Tonix’s lead candidate for CNS is TNX-102 SL (sublingual formulation of cyclobenzaprine), a non-opioid, centrally-acting analgesic that is currently in mid-Phase 3 development for the treatment of fibromyalgia. The Company reported positive data from a Phase 3 Relief study that met its pre-specified primary and secondary endpoints of pain reduction, improved sleep, reducing fatigue and overall improvement in symptoms and functions.

Image Source: Company
Tonix has randomized 50 percent of the participants in the second pivotal phase 3 RALLY study in fibromyalgia, which will be enrolling approximately 670 participants. The Company intends to announce the results from an interim analysis in Q3,2021, followed by topline results in Q4,2021. Tonix intends to submit a New Drug Application for the candidate to the U.S. Food and Drug Administration (FDA) in 2022, based on the outcome of its second phase 3 study.
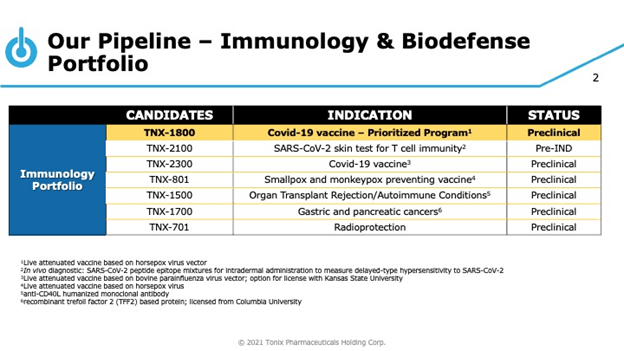
Image Source: Company
The Company is also awaiting the clearance for its Investigational New Drug (IND) from FDA for initiating a phase I clinical trial of TNX-1800, a single-administration COVID-19 vaccine candidate that elicits T cell immunity and is based on its horsepox virus vector, TNX-801. The candidate has demonstrated strong immune response to SARS-CoV-2 in non-human primates at low-doses, and the Company intends to announce results from an efficacy study of TNX-1800 in vaccinated non-human primates, that have been challenged with live SARS-CoV-2 in Q1,2021.
Tonix is also gearing up to file an IND and initiate clinical trials for TNX-2100, a diagnostic skin test in Q2, 2021. Furthermore, the Company is planning an IND application for TNX-1900 (intranasal potentiated oxytocin) in Q2,2021, besides initiating a Phase 2 study of TNX-1900 for the prophylactic treatment of chronic migraine in Q3,2021.
The Company has an extensive and diverse pipeline of candidates covering areas with high unmet medical needs such as Agitation in Alzheimers, Cocaine Addiction / Overdose, Migraine and Craniofacial pain, Prader- Willi Syndrome, Depression, PTSD and ADHD as part of its CNS development pipeline. Tonix’s Immunology Portfolio consists of vaccines against smallpox and monkeypox, organ transplant rejection and autoimmune conditions, gastric and pancreatic cancers and radioprotection.
Disclosure: I/we have no positions in any stocks mentioned, and no plans to initiate any positions within the next 72 hours.
Click here to please visit our detailed disclosure
References
https://hepionpharma.com/wp-content/uploads/2021/01/HEPA-Deck-Jan-2021.pdf
https://zomedica.com/wp-content/uploads/2021/03/March-2021-TRUFORMA-Brochure-1.pdf





No Comments