
17 May Can Minerva Neurosciences Roluperidone be a Turning Point in Schizophrenia Treatment?
A clinical-stage company Minerva Neurosciences, Inc. (NASDAQ: NERV) is developing an array of products for treating Central Nervous System (CNS) disorders, this biopharmaceutical company aims to deliver innovative solutions by harnessing the potential of serotonin receptors to combat disorders such as Schizophrenia and Parkinson’s disease.
The Company announced results from the 40-week open-label extension (OLE) of its phase 3 trial of roluperidone for the treatment of negative symptoms (NS) of schizophrenia. The results show a mean improvement in negative symptoms and improvement in emotional experience and social functioning.
Remy Luthringer, PhD Executive Chairman & Chief Executive Officer commented,
“I am delighted to announce that our one-year Phase 3 trial, now completed, supports our view that roluperidone, administered without concomitant treatment with antipsychotics, can potentially improve negative symptoms of schizophrenia and social functioning over the long term. We believe the data also suggest that roluperidone’s unique pharmacology and mechanism of action potentially help to maintain the stability of psychotic symptoms over the duration of treatment.”
Roluperidone (MIN-101) is a 5-HT2A antagonist, Sigma2 antagonist and α1A-adrenegic antagonist, three compounds that regulate cognition, mood, sleep and anxiety and is intended for the treatment of negative symptoms associated with Schizophrenia. The drug acts by blocking 5-HT2A, a serotonin receptor, which can lead to minimizing hallucinations, delusions, agitation and thought and movement disorders as well as antipsychotic treatment side effects. In addition, it also promotes slow wave sleep, improves memory and learning and increases calcium levels in the neurons in the brain.
Clinical Studies have shown the effect of roluperidone on Brain-Derived Neurotrophic Factor, which is responsible for neurogenesis, neuroplasticity, neuroprotection, synapse regulation, learning and memory. Besides Schizophrenia, the Company intends to pursue the therapeutic utility of roluperidone in other brain disorders that have negative symptoms such as Dementia, Autism spectrum disorders, Alzheimer’s disease, Parkinson’s disease and depression.
Minerva Neurosciences, Inc. (NASDAQ: NERV)
Market Cap: $ 125.17M; Current Share Price: 2.93 USD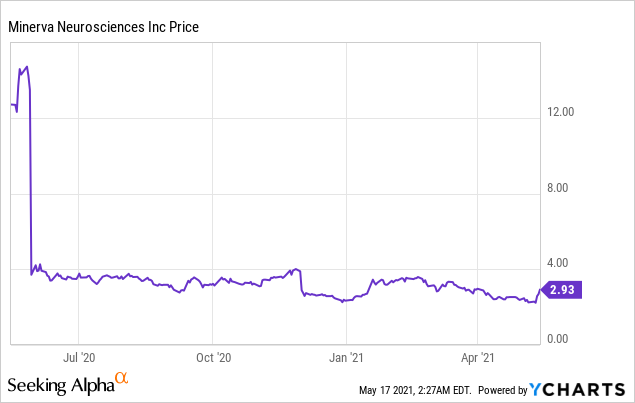
Data by YCharts
Industry
Schizophrenia is defined as a mental disorder in which there are disturbances in thought (delusions), perceptions (hallucinations), behavior (disjointed speech) and social interactions. It is one of the leading causes of disability worldwide. In the U.S alone mental and behavioral disorders account for 13.6 percent of total DALY’s (Disability-adjusted life years) according to the statistics released by National Institute of Mental Health.
Schizophrenia is characterized by symptoms such as Failure to distinguish between perception and reality, Hallucinations which are visual (Seeing people), Auditory (Hearing voices), or related to taste, touch and the sense of smell, Suffering from delusions related to being persecuted or being victimized, Suffering from sexual and religious delusions, Speech which does not follow any logic or incoherent thoughts, Exhibiting social withdrawal, reticence, lack of self-care, emotionally distant and lack of motivation.
The treatment for schizophrenia is life-long, even when the symptoms have reduced. Anti-Psychotic drugs that can control dopamine and glutamate are the most preferred course of medication. These are usually combined with psychosocial therapy (family therapy, individual counselling, rehabilitation and employment support) and other drugs such as antidepressants and anti-anxiety medication.
Research shows that a whopping 86.19% of people experience at least one of the above-mentioned side effects prompting them to stop taking medication. Only 42.5% of the patients reported total adherence to the medication. These side effects prevent people from taking their medication diligently, thereby leading to worsening of symptoms overtime.
Company
The company’s drug candidate for Parkinson’s, MIN-301, is a neuregulin-1β1 protein that can slow the onset and repair the tissue damage caused by Parkinson’s. It has shown positive effects in cognition and restoration of motor function in clinical trials. The drug seeks to overcome the limitations of existing medications by delivering improved performance on parameters such as faster action, prolonged effect, minimal side effects and reduced cognitive impairment.
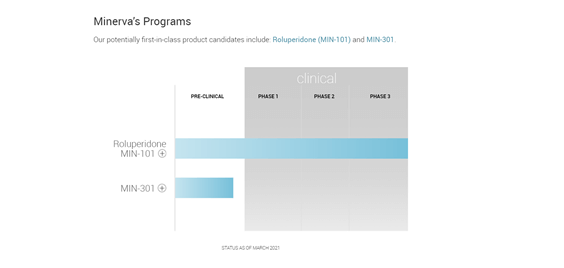
Image Source: Company
Minerva has previously developed Seltorexant through a licensing agreement with Janssen Pharmaceuticals N.V., a Johnson & Johnson company (NYSE: JNJ). The candidate was under development for insomnia disorder and adjunctive treatment of Major Depressive Disorder (MDD). In December 2020, the Company opted out of the joint agreement and is eligible to collect royalties in the mid-single digits on potential sales of seltorexant worldwide in certain indications in the future. The Company does not have any financial obligations to Janssen and will not be playing any part in the future development of seltorexant.
Furthermore, in January 2021, Royalty Pharma acquired the Company’s royalty interest in seltorexant for an upfront payment of $60 million and up to $95 million in additional milestone payment, which are dependent on achievement of certain clinical, regulatory and commercialization milestones. The candidate is currently undergoing Phase 3 development for treatment of major depressive disorder (MDD) with insomnia symptoms.
Key Takeaways
Disclosure: I/we have no positions in any stocks mentioned, and no plans to initiate any positions within the next 72 hours.
Click here to please visit our detailed disclosure
References
https://www.merriam-webster.com/dictionary/schizophrenia
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1914490/
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3342101/
https://www.ncbi.nlm.nih.gov/pubmed/29241357
https://www.minervaneurosciences.com/programs/
https://finance.yahoo.com/news/minerva-neurosciences-announces-results-phase-201500088.html
https://www.fortunebusinessinsights.com/industry-reports/schizophrenia-drugs-market-101531





No Comments