
21 Oct Oyster Point Pharma Hits the Bulls EYE!
Oyster Point Pharma, Inc. (NASDAQ: OYST) is a commercial-stage biopharmaceutical company creating first-in-class therapies for the treatment of ophthalmic conditions. The Company’s lead product TYRVAYA, (varenicline solution) Nasal Spray 0.03 mg (formerly referred to as OC-01), has become the first and only FDA-approved nasal spray, intended for the treatment of dry eye disease.
Oyster Point Pharma, Inc. (NASDAQ: OYST)
Market Cap: $331.17M; Current Share Price: 12.73 USD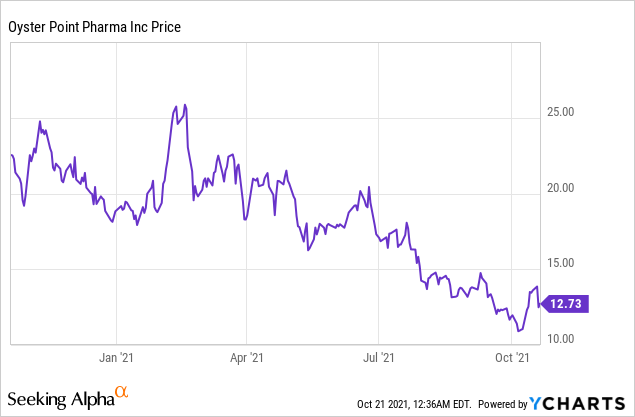
Data by YCharts
Commenting on the approval, Jeffrey Nau, Ph.D., MMS, president and CEO of Oyster Point Pharma stated
“The approval of TYRVAYA Nasal Spray marks a milestone for patients and eye care professionals by providing a new drug treatment option for the signs and symptoms of dry eye disease with a differentiated route of administration that is believed to leverage a nerve pathway that can be accessed within the nose. In any therapeutic area, it’s always an exciting moment when you follow the science and develop a truly innovative pharmaceutical treatment option for patients that addresses an important unmet medical need. In conjunction with the FDA, it has been an honor to work alongside my colleagues at Oyster Point to bring TYRVAYA Nasal Spray to the dry eye disease community. We look forward to making TYRVAYA Nasal Spray available to eye care professionals and their patients.“
TRYVAYA is a highly selective cholinergic agonist, meant to be administered twice daily into each nostril to activate basal tear production. The drug was studied in multiple clinical trials namely ONSET-1, ONSET-2, and MYSTIC in over 1,000 patients with mild, moderate or severe dry eye disease. TRYVAYA has demonstrated statistically significant improvements in tear film production, with more than 52 percent achieving ≥10 mm increase in Schirmer’s score in ONSET 1 study and 47 percent achieving ≥10 mm increase in Schirmer’s score from baseline in the ONSET-2 study, compared to 14% and 28% of vehicle-treated patients in the ONSET-1 study and the ONSET-2 study, respectively at Week 4 (p<0.01 in both studies).
The drug will be available with a prescription from November 2021.

Image Source: Company
Strength
The Company is leveraging its expertise in nicotinic acetylcholine receptor (nAChR) domain expertise to create a diverse and extensive pipeline of candidates such as neurotrophic keratopathy, dry eye associated with contact lens intolerance, and ocular surface preparation for refractive surgeries among others.
Oyster Pharma has developed a proprietary gene therapy namely, Enriched Tear Film (ETF™) for the treatment of ocular surface diseases, by delivering target genes to diseased cells using adeno-associated virus (AAV) vectors. The Company’s approach is to work on the lacrimal gland, which is capable of producing selected proteins in the body’s own natural tear film to treat specific ocular surface diseases.
Preclinical studies show that OC-01 (varenicline solution) nasal spray can modulate proteins, peptides, hormones, enzymes and cytokines in the lacrimal gland and can be leveraged to treat numerous disorders. The Company has entered into a research collaboration to develop treatments for bacterial infections in ophthalmology using bacteriophages, viruses found in the natural environment.
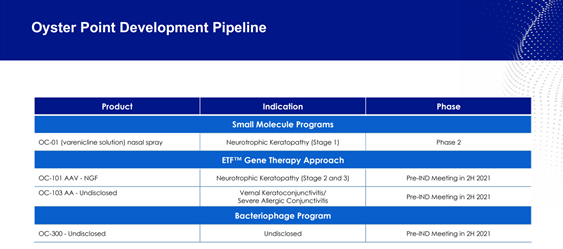
Image Source: Company
Oyster’s pipeline consists of OC-1, intended for the treatment of Neurotrophic Keratopathy, which is currently enrolling patients in a Phase 2 trial. In addition, the company is exploring the potential of its proprietary Enriched Tear Film (ETF™) Gene Therapy through development of OC-101 AAV -NGF (Neurotrophic Keratopathy) and OC-103 AAV-X (Vernal Kerato-conjunctivitis/Severe Allergic Conjunctivitis that are currently under preclinical evaluation. The Company intends to schedule a pre-IND meeting with the FDA in the second half of 2021, along with a meeting for OC-300, its Bacteriophage candidate.
Weakness
In August 2021, the Company secured a $125 million term loan credit facility with OrbiMed Royalty & Credit Opportunities III, LP. The loan amount was to be disbursed in three separate tranches, with $45 million being paid upfront, followed by a second tranche of $50 million to be received upon FDA approval. The final tranche of $30 million is payable on meeting a recurring sales threshold. However, the second tranche is only payable if the approved label includes eye dryness score data from the clinical trials conducted by the Company.
During clinical trials, patients are made to evaluate their level of eye dryness discomfort in both clinical as well as controlled adverse environments, leading to the tabulation of an eye dryness score. The drug achieved its objectives in the clinical settings, however due to the COVID-19 pandemic some of the trial patients were unable to visit sites, or sites were unable to accommodate patients, which resulted in data that was not statistically significant. The FDA-approved label therefore does not include this data, which may result in the Company failing to secure the second tranche and subsequently the third tranche as well.
Oyster is negotiating with OrbiMed for a waiver of the eye dryness score requirement. In case, the Company fails to secure the loan amount, it may need to raise capital through other forms such as a share offering, leading to dilution of shareholder value.
The Company had cash and cash equivalents of $184 million as of Sept. 30, 2021, however the commercial launch of TYRVAYA may need more funds.
Opportunity
Dry Eye Syndrome is characterized by the inability to produce sufficient amount or quality of tears which leads to lack of lubrication and nourishment to the surface of the eye and manifests as chronic eye irritation, inflammation and damage to the surface. There are over 16 million diagnosed cases of dry eye syndrome in the U.S alone.
Dry Eye leads to discomfort and pain and can greatly impact the quality of life by making it difficult to perform everyday functions such as reading, driving, using computers and performing daily activities. In the U.S the financial implications of dry eye management was estimated to be US$ 11,302 per patient and US$ 55 billion overall according to a report.
Image Source: aoa.org
There is no cure for dry eye disease currently and treatment options include topical medication, punctal plugs, practitioner visits, and nutritional supplements, these options are highly inadequate and hence the need for novel solutions. The prevalence of dry eye ranged from 5 to 30 % of individuals aged over 50 according to an estimate by The Epidemiology Subcommittee of the 2007 DEWS. According to an estimate by the National Health and Wellness Survey, over 16.4 million adults (6.8 percent of the population) in the U.S have been diagnosed with Dry Eye Disease.
A report by Transparency Market Research projects that the industry will be worth $7.78 billion by 2025, growing at a CAGR of 4.5%.A rising geriatric population, increased screen exposure, diabetes, environmental factors such as pollution and refractive surgeries are leading to a rise in the incidences of dry eyes, while the lack of adequate therapeutic options, expensive medication along with lack of patient adherence is a challenge for the growth of the market.
Threats
Clinical Trials are fraught with risk and uncertainty. There is a possibility that the candidates in the Company’s developmental pipeline may not be able to meet their clinical endpoints in trials. However, a diverse pipeline will help mitigate the risk in case of adverse results or the failure to meet endpoints in any of its ongoing trials. The success of its clinical trials will help the Company advance its pipeline but it should also be prepared to face any setbacks, in case its ongoing trials fail to meet their endpoints.
Key Takeaways
Disclosure: I/we have no positions in any stocks mentioned, and no plans to initiate any positions within the next 72 hours.
Click here to please visit our detailed disclosure
References
https://www.aoa.org/healthy-eyes/eye-and-vision-conditions/dry-eye?sso=y
https://www.uptodate.com/contents/dry-eye-disease
https://investors.oysterpointrx.com/static-files/69b69a85-3566-44f8-8c01-cb0a84dee1dc





No Comments