
19 Oct All you need to know about NRx Pharmaceuticals!
NRx Pharmaceuticals, Inc. (NASDAQ: NRXP) is a clinical-stage biopharmaceutical company focused on creating novel therapeutics that use molecules to treat medical conditions with no approved cure. The Company’s lead product candidate is ZYESAMI™ (AVIPTADIL®) ZYESAMI™ (Aviptadil acetate), a synthetic Vasoactive Intestinal Peptide (VIP), which has been granted fast track designation by the U.S. Food and Drug Administration (FDA) for the treatment of respiratory failure in critical COVID-19 patients. In addition, the candidate has been provided an Expanded Access Protocol by the FDA, signifying that the candidate has the potential to treat a serious or life-threatening condition.
In May 2021, the Company had submitted an Emergency Use Authorization (EUA) in the U.S.
NRx Pharmaceuticals, Inc. (NASDAQ: NRXP)
Market Cap: $546.88M; Current Share Price: 10.17 USD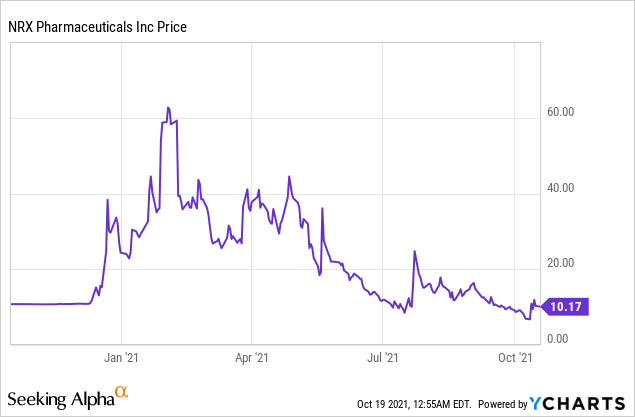
Data by YCharts
Strengths
ZYESAMI™, which was evaluated in a Phase 3 trial has demonstrated to be more effective than a placebo in treating patients suffering from Critical COVID-19 with respiratory failure. ZYESAMI™ is essentially a synthetic form of the human Vasoactive Intestinal Peptide (VIP) that is produced by the body and aids in protecting the cells against inflammatory conditions. Data suggests that VIP binds to receptors of Alveolar Type II cells, which the SARS-CoV-2 virus also bind to. VIP is not only capable of inhibiting the replication of SARS-CoV2 virus, but also prevents cytopathy and upregulates the production of surfactant, thereby protecting cells and pulmonary epithelium by blocking cytokines.

Image Source: Company
The candidate has shown to reduce hospital stay and improve the chances of survival, however with a common adverse event, namely diarrhea. The results from the COVID-AIV trial, which had enrolled 196 patients who were administered ZYESAMI™ (Aviptadil), through intravenous infusion, achieved its primary endpoints of improvement in blood oxygenation and mortality.
The Company is also evaluating ZYESAMI™, in a Phase 3 trial, in patients who have severe COVID-19 but have not yet developed respiratory failure. The aim is to study the efficacy of the candidate in reducing the progression of COVID-19 to a critical-stage, improving survival rates and shortening hospital stay.
Furthermore, the Biomedical Advanced Research and Development Authority (BARDA) of the US Department of Health and Human Services has sponsored a Phase 2 study, namely I-SPY, of inhaled ZYESAMI™ in treating patients with Critical COVID-19 who have already developed respiratory failure, to gauge its efficacy in increasing the survival rates and prevention of respiratory failure at 90 days compared to treatment with placebo or other drugs. NRx is also evaluating ZYESAMI™ in a Phase 3 ACTIV3b/TESICO trial.
The Company has entered into an agreement with the Government of Israel for the exclusive worldwide rights to BriLife™ COVID vaccine developed by the Israel Institute for Biological Research.
Weakness
The Company recently announced that Relief Therapeutics Holding AG and Relief Therapeutics International SA has filed a lawsuit against the Company, claiming failure to honor its obligations under the collaboration agreement entered between the companies in September 2020. The lawsuit is seeking damages for breach of contract.
As part of the collaboration agreement, Relief was to take care of the funding, while NRx focused on the research and development for aviptadil. However, as per NRx, Relief stopped funding the project in January 2021 after investing only $10.9 million, stating that the drug had failed and “missed the pandemic”. Consequently, NRx continued to develop the drug and incurred more than $25 million for the research and development of the drug.
Relief has also alleged that NRx failed to divulge necessary information on how the funds were intended to be spent, which the Company has rebuffed. The Company claims to have provided all necessary information to Relief and believes that the claims are baseless and without merit.
The Company intends to defend the case vigorously and will assert significant counterclaims against Relief, at the appropriate time, as per its filing of a Form 424B3.
Opportunity
According to the World Health Organization (WHO), Coronavirus (CoV) is a family of viruses that cause illnesses ranging from common cold to lethal diseases such as the Middle East Respiratory Syndrome (MERS-CoV), Severe Acute Respiratory Syndrome (SARS-CoV) and a new strain of CoV named, novel coronavirus (nCoV). The virus is Zoonotic as it is transmitted from animals to humans, with the presence of many strains that have not yet infected humans. The infection manifests in the form of respiratory symptoms, cough, fever, shortness of breath and can take the form of pneumonia, severe acute respiratory syndrome, kidney failure and even death.
WHO had identified a new type of coronavirus, namely 2019 novel coronavirus (2019-nCoV) in China in early January 2020, which has so far infected 240,061,454, including 4,887,600 deaths, according to the WHO Coronavirus dashboard. The disease which originated from the Chinese city of Wuhan, has spread to 168 countries, areas and territories. According to the US Centers for Disease Control and Prevention (CDC), there are 44,801,768 known cases and 722,212 people have died so far in the U.S alone (as of October 17, 2021).
There is no definite cure for treatment or eradication of CoV, with treatment being limited to alleviation of symptoms, and providing support and care to patients. Scientists have been able to sequence the virus genetic code; spurring organizations around the world into action.
The COVID-19 pandemic is likely to become endemic, similar to the seasonal FLU and may lead to over 300 thousand Critical cases per year, in addition to the already existing ARDS cases that come close to 200 thousand. Though vaccination has helped improve the situation, the rising mutations and breakthrough infections add to the uncertainty of vaccine efficacy. The need of the hour is a treatment that can help prevent and manage respiratory failures that lead to mortality.
Threats
Clinical Trials are fraught with risk and uncertainty. There is a possibility that the candidates in the Company’s developmental pipeline may not be able to meet their clinical endpoints in trials. However, a diverse pipeline will help mitigate the risk in case of adverse results or the failure to meet endpoints in any of its ongoing trials. The success of its clinical trials will help the Company advance its pipeline but it should also be prepared to face any setbacks, in case its ongoing trials fail to meet their endpoints.
Pipeline
NRx plans to explore the multiple applications for ZYESAMI and is looking beyond COVID-19, for instance the Company is also targeting ARDS, Sarcoidosis and other indications such as checkpoint inhibitor Pneumonitis, COPD, lung transplant and amniotic fluid embolus among others.
The Company’s pipeline consists of NRX-100/NRX-101™, intended for the treatment of Severe Bipolar Depression in patients with Acute suicidal ideation, that is currently undergoing Phase 2 trials , NRX-101™ in Severe Bipolar Depression in patients with Acute suicidal ideation, which is currently in Phase 2/b/3 trials, NRX-100™, intended for the treatment of Acute Suicidality Ideation and Behavior (ASIB) in Bipolar Depression, currently undergoing Phase 3 trials and NRX-101™ in Moderate Bipolar Depression with Suicidal Ideation, undergoing Phase 2/3 trial.

Image Source: Company
NRX-101 has been granted a breakthrough designation and Special Protocol Agreement by the U.S FDA, in addition to Biomarker Letter of Support, and Fast Track Designation.
Key Takeaways
- The Company has recently submitted a revised Investigational New Drug (IND) module with details pertaining to the manufacturing of ZYESAMI, including information about Nephron Pharmaceuticals supplying the drug on a commercial scale. NRx also disclosed that an inspection of its manufacturing facility by European authorities has been completed with no adverse findings. This would help the Company file approval requests in Europe and U.K. The drug has already been approved in Georgia.
- NRx intends to announce data from the BARDA sponsored I-SPY trial in the second half of 2021, along with data from the Phase 2b & Phase 3 COVID Vaccine Study. The Company intends to announce data from the NIH-sponsored ZYESAMI trial in the first of 2022, along with filing for an NDA for the candidate, with a potential approval in 2022.
- The Company has plans to enter into a commercial services agreement with a third party upon receipt of a EUA in the U.S. Furthermore, the Company has entered into an agreement with Cardinal Health Specialty Distribution & 3PL to manage the necessary logistics upon receipt of the authorization.
- The Company is likely to announce the top-line results from NRX-101, with a potential NDA filing expected in the second half of 2022. NRx is targeting a potential $5 billion total addressable market in suicidal depression and PTSD.
- The Company is helmed by an experienced management team, with a vast experience in drug development. The Founder, Chairman and CEO of the Company, Jonathan C. Javitt, MD, has successfully participated in 6 drug and device launches at Merck, Allergan, Eyetech. The leadership team has many more such accomplished individuals who bring a lot of value to the table.
- The Company holds the composition of matter patent protection for NRX-101, encompassing 5 patent families and more than 30 issued and 60+ pending patents in the U.S, European Union, Canada, Japan, and Australia.
Disclosure: I/we have no positions in any stocks mentioned, and no plans to initiate any positions within the next 72 hours.
Click here to please visit our detailed disclosure
References
https://www.nrxpharma.com/zyesami/
https://covid.cdc.gov/covid-data-tracker/#cases_totalcases
https://www.streetinsider.com/SEC+Filings/Form+424B3+NRX+Pharmaceuticals%2C/19064437.html





No Comments