30 Apr Can Humanigen’s lenzilumab Prove to be a Gamechanger for the Company?
Humanigen, Inc. (NASDAQ: HGEN) is a clinical-stage biopharmaceutical Company that is focused on creating a portfolio of monoclonal antibodies targeting immunology and immune-oncology disorders. The Company’s lead product candidate is lenzilumab, an anti-human granulocyte-macrophage colony-stimulating factor (“GM-CSF”) monoclonal antibody, formulated using the Company’s proprietary Humaneered® technology.
A Differentiated Approach
The Company is leveraging its proprietary and patented Humaneered technology platform, to convert existing murine antibodies into engineered, high-affinity human antibodies, which can target chronic conditions. Humanigen has acquired antibody research candidates from academic institutions and through in-house development and has transformed them through the application of its Humaneered technology.
A major differentiating factor of the Company’s antibodies is that they are closer to human antibodies than chimeric or conventionally humanized antibodies on one hand and have low immunogenicity on the other, an improvement over the conventional approach.
Humanigen, Inc. (NASDAQ: HGEN)
Market Cap: $1.09B; Current Share Price: 18.55 USD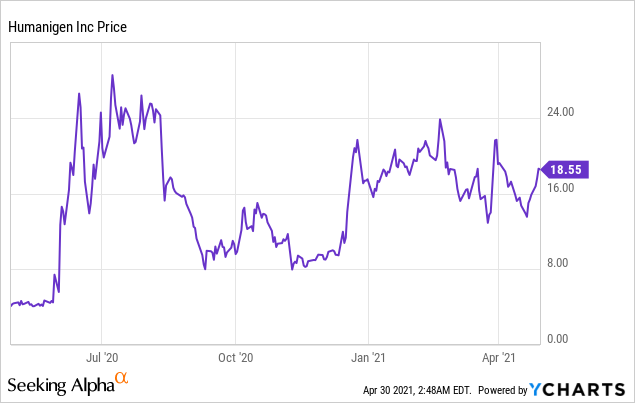
Data by YCharts
Industry
According to the World Health Organization (WHO), Coronavirus (CoV) is a family of viruses that cause illnesses ranging from common cold to lethal diseases such as the Middle East Respiratory Syndrome (MERS-CoV), Severe Acute Respiratory Syndrome (SARS-CoV) and a new strain of CoV named, novel coronavirus (nCoV). The virus is Zoonotic as it is transmitted from animals to humans, with the presence of many strains that have not yet infected humans. The infection manifests in the form of respiratory symptoms, cough, fever, shortness of breath and can take the form of pneumonia, severe acute respiratory syndrome, kidney failure and even death.
There is no definite cure for treatment or eradication of CoV, with treatment being limited to alleviation of symptoms, and providing support and care to patients. Scientists have been able to sequence the virus genetic code; spurring organizations around the world into action.
Various studies have pointed to the fact that critically ill patients demonstrate a high concentration of pro-inflammatory cytokines compared to those who are moderately ill, indicating a poor prognosis in cases where the levels are extremely high. Cytokine Storm is a broad term that encompasses several life-threatening systemic inflammatory syndromes that are marked by immune-cell hyperactivation and can be caused due to multiple reasons such as cancers, monogenic disorders, autoimmune disorders and pathogens. Cytokine storms cause the body to attack its own cells and tissues instead of fighting with an invading virus.
Other studies show that increased levels of pro-inflammatory plasma cytokines is critical to the severity of the disease. Though the exact reason for the response is still unknown, the role of interleukin-6 (I6) amplifier is being evaluated by scientists to gain a deeper understanding of the phenomenon.
Lenzilumab – A Winner in the Making!
Lenzilumab has demonstrated the potential to neutralize GM-CS, a critical cytokine in the hyperinflammatory cascade, by binding and neutralizing GM-CSF in animal models. The candidate has been evaluated in a phase 3 registrational clinical trial for treatment of COVID-19 pneumonia, and has shown that it improves survival without the need for invasive mechanical ventilation (IMV), by nearly 54 percent. The candidate also registered a better clinical improvement when compared to other treatments including steroids and/or remdesivir.
Cameron Durrant, MD, MBA, Chief Executive Officer, Humanigen commented,
“The results from our Phase 3 clinical trial with lenzilumab treatment were associated with better outcomes in hospitalized hypoxic COVID-19 patients who had not yet progressed to the point of requiring IMV. Additionally, the trial incorporated a diverse population with various comorbidities, most commonly a body mass index above 30, which is representative of a real-world, high-risk population. Our next step is to submit an application for Emergency Use Authorization (EUA) to the Food and Drug Administration (FDA) as soon as possible. We are also sharing these results with US governmental agencies and other authorities worldwide.”
The phase 3 study is a randomized, double-blind, placebo-controlled, multi-center study that had the primary objective of studying Lenzilumab’s potential to reduce the immune-mediated cytokine release syndrome (CRS) and improve ventilator-free survival. The trial was conducted in over 520 patients in 29 sites in the US and Brazil, all above the age of 18 years. The secondary endpoints measured, through 28 days, parameters such as ventilator-free days, duration of ICU stay, incidence of invasive mechanical ventilation, extracorporeal membrane oxygenation (ECMO), and/or death.

Image Source: Company
The Company is now planning to file an Emergency Use Authorization with the FDA, making it the first antibody treatment for hospitalized patients, besides being the first treatment for COVID-19 related cytokine storm.
The candidate is also being evaluated in the NIAID-sponsored ACTIV-5/Big Effect Trial in hospitalized patients with COVID-19. The Company had previously entered into a partnership with National Institute of Allergy and Infectious Diseases as part of its strategic priority to advance therapeutic candidates with novel mechanisms of action for treatment of COVID-19.
A pipeline focused on Multiple Opportunities
Humanigen has also reported positive data from the Phase 1b portion of its study in large B-cell lymphoma (DLBCL) that is evaluating the safety and efficacy of lenzilumab in patients treated with CAR-T with relapsed or refractory DLBCL. In addition, the Company is also collaborating with IMPACT, to initiate a potential randomized, placebo controlled, double-blind, Phase II/III study for evaluating the safety and efficacy of lenzilumab in reducing non-relapse mortality at six months, in patients at high or intermediate risk for steroid refractory acute GvHD.
The Company’s pipeline also consists of a phase 2 evaluation of Chronic myelomonocytic leukemia (CMML) with an undisclosed partner and a phase 1 trial in Solid Tumors (Glioblastoma Multiforme) that is being developed in Australia in association with the QIMR Berghofer Medical Research Institute and Olivia Newton-John Cancer Research Institute.

Image Source: Company
Key Takeaways
Humanigen’s differentiated approach of focusing on reduction of cytokine storms has the potential to reduce the need for ventilation in severely ill Covid-19 patients by 54 percent, which could be potentially life saving for many. While the U.S has carried out vaccinations of its elderly population, those most at risk of needing ventilation, on a war-footing, the Company has plenty of opportunities to expand its offerings to other geographic locations such as countries which still haven’t fully vaccinated their population.
Clinical Trials are fraught with risk and uncertainty. Humanigen is currently focused on Lenzilumab and is working on developing a diverse pipeline of candidates such as Ifabotuzumab, which will help mitigate the risk in case of adverse results or the failure to meet endpoints in any of its ongoing trials. The success of its clinical trials will help the Company advance its pipeline but it should also be prepared to face any setbacks, in case its ongoing trials fail to meet their endpoints.
Disclosure: I/we have no positions in any stocks mentioned, and no plans to initiate any positions within the next 72 hours.
Click here to please visit our detailed disclosure
References





No Comments