
06 Nov Cyclerion Therapeutics Praliciguat Fails in Two Crucial Clinical Trials
Cyclerion Therapeutics (NASDAQ: CYCN), a clinical-stage Company, announced the failure of its drug candidate praliciguat in meeting the primary endpoints in two separate clinical studies, in diabetic nephropathy (DN) and heart failure with preserved ejection fraction (HFpEF). Praliciguat is a soluble guanylate cyclase (sGC) stimulator indicated for the treatment of cardiometabolic diseases, which was granted a Fast Track Designation by the U.S FDA for the treatment of HFpEF. In preclinical studies, the drug demonstrated extensive distribution to adipose tissue, kidney, heart and liver.
As per a Company statement, the study did not achieve statistical significance on its primary endpoint of reduction in albuminuria from baseline as compared to placebo, measured by urine albumin creatinine ratio (UACR). However an improvement was observed in the total intention-to-treat (ITT) study population, besides improvements in secondary vascular and metabolic measures associated with cardiovascular risk and kidney disease progression, including blood pressure, cholesterol and HbA1c levels. The study also revealed a favourable pharmokinectic profile of praliciguat, with the drug being generally well tolerated and safe.
Ian de Boer, M.D., M.S., Professor, Division of Nephrology, Adjunct Professor, Epidemiology and Associate Director, Kidney Research Institute at University of Washington commented
“I am encouraged by the estimated reduction in albuminuria of 15% or more, compared with placebo, on top of current standard of care. This molecule modifies pathways that are complementary to those targeted by usual care, and it warrants further investigation as a potential treatment for patients with diabetic kidney disease”.
The Company intends to out-license praliciguat for late-stage development and commercialization in DN, HFpEF and other cardio metabolic indications, after the completion of its phase 2 studies in DN and HFpEF.
Mark Currie, Ph.D., president and chief scientific officer at Cyclerion stated
“We believe praliciguat has the potential to be a first-in-category treatment for patients with diabetic nephropathy. We look forward to sharing the data with prospective partners.”
Cyclerion is now planning to focus on its STRONG SCD, a phase 2 study of olinciguat, a sGC stimulant similar to praliciguat, being developed for the treatment of sickle cell disease, the results of which are expected to be announced in mid-2020. In addition the Company is also engaged in a Phase I clinical trial named IW-6463 a central nervous system-penetrant sGC stimulator, the results of which are due in Q4 2019.
The Company has announced its plan to reduce its monthly cash expenses by 25 percent, focus on creating near-term value-creation opportunities such as out-licensing and partnering agreement and layoff 30 of its 135 staffers.
Cyclerion presently has nearly $125 million in cash, cash equivalents and restricted cash as of September 30, 2019, which it believes will be sufficient to fund its ongoing trials and operations until Q1, 2021.
Cyclerion Therapeutics (NASDAQ: CYCN)
Market Cap: $61.52M; Current Share Price: 2.30 USD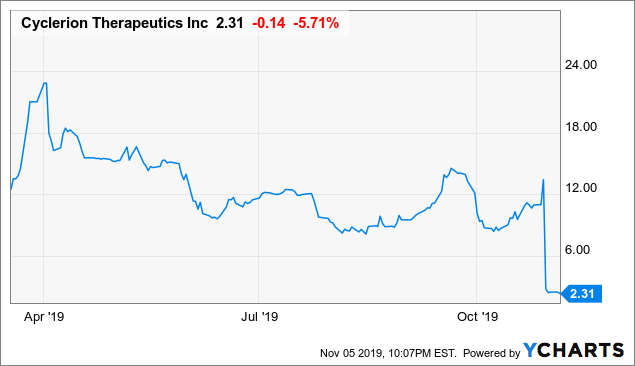
Data by YCharts
Industry
Diabetes is a silent killer which is slowly reaching endemic proportions in developed as well as developing countries. The rise in obesity, sedentary lifestyles, poor dietary choices, stress and lack of exercise will see the number of incidences of Diabetes rise in the future. According to the International Diabetes Federation, the number of people living with diabetes will rise to 629 million by 2045, from 425 million adults (20-79 years) in 2017. In the U.S alone the people living with diabetes are set to increase from 58 million in 2017 to 67 million by 2045.
The increased awareness about the long-term complications caused by diabetes, along with the rise in co-morbidities such as cardiovascular, renal and neuropathic complications is set to propel the growth in the segment from $26.8 billion in 2016 to around $64 billion by 2026 according to a research by GlobalData. In spite of the market being dominated by generic drugs, such as metformin and similar drugs being in developmental stage, the market is projected to achieve a CAGR of 8.4% as per the same report.

Image Source: Company
It is estimated that around 25 percent of the people with diabetes eventually develop kidney ailments such as Diabetic Nephropathy and Focal Segmental glomerulosclerosis (FSGS). These diseases hamper the kidney’s ability to function by preventing proper elimination of waste products and fluids from the body. The disease initially manifests in the form of unregulated blood pressure, protein in the urine, swelling, and shortness of breath, nausea, itching and fatigue and can lead to end-stage renal failure if left untreated.
Heart failure with preserved ejection fraction (HFpEF) is a form of heart failure in which the blood ejected from the left ventricle (ejection fraction) is greater than 50 percent, which eventually leads to heart failure. This condition is caused by co-morbidities such as hypertension, diabetes, smoking or sleep apnea. According to an estimate by the American Heart Association, over 6.5 million people suffer from heart failure, with the number likely to reach 8 million by 2030. There are more than 960,000 new diagnoses of HF every year, according to data from the National Health and Nutrition Examination Survey.
Company
Cyclerion Therapeutics is exploring the potential of the NO-cGMP pathway, by developing next-generation soluble guanylate cyclase (sGC) stimulators to address debilitating orphan diseases. Cyclerion’s pipeline includes five sGC stimulator programs indicated for the treatment of Sickle cell disease, DN, HFpEF, CNS, Liver and Lung orphan disease conditions.

Image Source: Company
The Company is targeting the nitric-oxide signaling, a crucial component of real-time regulation of various biological systems. Soluble guanylate cyclase (sGC) play an important role in regulating Nitric oxide signaling, and is present in tissues throughout the body, including in the vasculature, kidney, brain, lung, heart, liver, adipose and skeletal muscle. Nitric Oxide produced locally percolates into target cells and binds to sG, which in turn catalyzes the conversion of guanosine – 5’ – triphosphate (GTP) to cyclic guanosine monophosphate (cGMP), effecting a marked improvement in the secondary signaling molecule.

Image Source: Company
Cyclerion’s strategy is to design novel small molecule sGC stimulators, which are capable of increasing nitric oxide signalling in disease-relevant tissues, thereby resulting in improved production of cGMP. The Company has sights on extending the application of this technology in developing therapeutics for diseases associated with inflammation, fibrosis or metabolic dysregulation.

Image Source: Company
The Company was spun out of Ironwood Pharmaceuticals (NASDAQ: IRWD) in April 2019.
Risk Assessment
- The failure of its clinical trial in HFpEF has promoted the Company to stop further development of praliciguat in heart failure. However, since the patients participating in the diabetic nephropathy trial exhibited a reduction in their HbA1c levels and overall improvement in the total intention-to-treat (ITT) study population, the Company is planning to out-license the drug for further development for the treatment of Diabetic Nephropathy.
- Cyclerion is expecting data from its phase 2 STRONG –SCD study of olinciguat, a vascular sGC stimulator, intended for the treatment of sickle cell disease (SCD) in mid-2020. The positive results from this trail will help the Company bounce back from the current setback and will help raise additional capital to fund its other clinical trials and run its operations.
- The Company is reducing its staff of 135 members by 30, thereby cutting back its monthly expenses by at least 25 percent. This will reduce its cash burn and keep it afloat until 2021, with the help of the $125 million in cash, cash equivalents and restricted cash that the Company had as of September 30, 2019.
- Cyclerion might find it challenging to find interested parties to out-license praliciguat, even in case of diabetic nephropathy, owing to the not so encouraging results from the clinical trials. The Company does not have commercialized products yet in the market and will be relying heavily on positive data from the impending IW-6463 Phase 1 study results expected to be announced in 4Q19.
Disclosure: I/we have no positions in any stocks mentioned, and no plans to initiate any positions within the next 72 hours.
Click here to please visit our detailed disclosure
Source:
https://www.mayoclinic.org/diseases-conditions/diabetic-nephropathy/symptoms-causes/syc-20354556





No Comments