
08 Oct Is it the end of the road for Ardelyx Inc?
Ardelyx, Inc. (NASDAQ: ARDX) lead candidate Tenapanor, is a targeted, first-in-class, oral, small molecule intended for the control of serum phosphorus in adult patients with chronic kidney disease (CKD) on dialysis. The drug has the potential to become a foundational therapy for hyperphosphatemia and enable effective control of elevated phosphate in the blood. Tenapanor’s unique mechanism of action inhibits the sodium/hydrogen exchanger 3 (NHE3), by acting locally in the gut, resulting in decreased phosphate absorption through its effect on the paracellular pathway.
Ardelyx, Inc. (NASDAQ: ARDX)
Market Cap: $130.96M; Current Share Price: 1.27 USD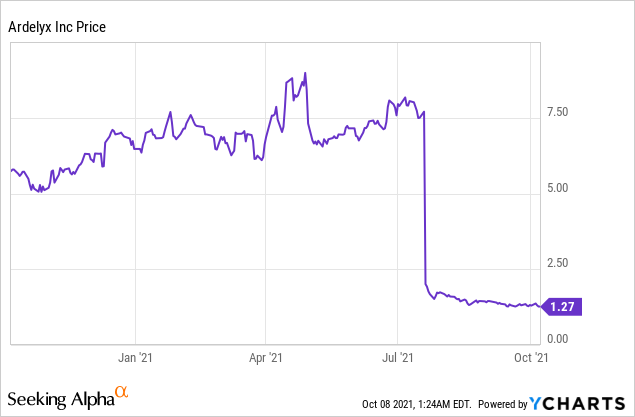
Data by YCharts
However, in July,2021 the Company received a Complete Response Letter (CRL) related to the New Drug Application (NDA) for tenapanor. The CRL states that the magnitude of the treatment effect as “small and of unclear clinical significance.” The FDA has requested Ardelyx to conduct an additional well-controlled trial that can demonstrate significant treatment effects of serum phosphorus. The Company intends to request for a Type A meeting with the FDA to determine the future course of action for the approval of tenapanor.
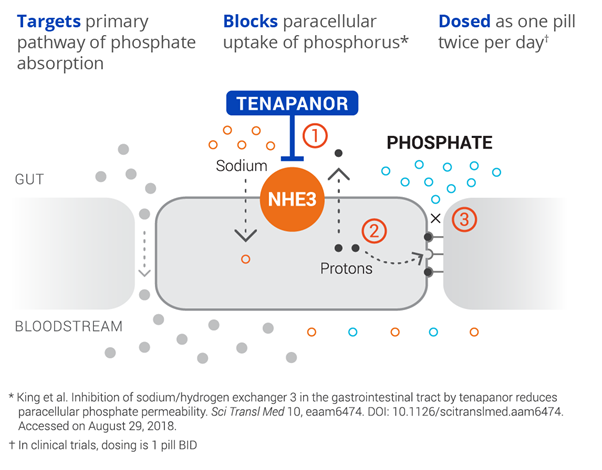
Image Source: Company
The Company has conducted three Phase 3 clinical trials, two monotherapy trials (BLOCK and PHREEDOM) and one dual mechanism trial (AMPLIFY), which demonstrate that Tenapanor can reduce phosphorus if taken twice a day, as both a monotherapy and as part of a dual-mechanism approach. The Company submitted a New Drug Application to the U.S. FDA in June, 2020 and was granted a Prescription Drug User Fee Act (PDUFA) of April 29, 2021 with a potential approval and eventual launch by Q3,2021.
Phase 3 trial results demonstrate that adult patients with CKD-HP using Tenapanor had a smaller percentage of deaths and hospitalizations compared to those on Sevelamer. In addition, treatment with Tenapanor was found to reduce serum phosphorus concentrations and mean serum phosphorus from 7.7mg/dL to 5.1mg/dL.
A Missed Opportunity?
Tenapanor was expected to be a potential blockbuster drug with several hundred million dollars in annual sales as it offered a reduced daily pill burden, when compared to phosphate binder therapies such as sevelamer. The news was surprising as the Company had also conducted comprehensive label discussions and filed additional response data with the FDA in April, besides meeting key primary and secondary endpoints in phase 3 trials. The extension of the April ,2021 PDUFA date was attributed to making certain amends to the NDA, with no mention of any concerns about the size of “the treatment effect or clinical relevance”, especially since the FDA actively helped Ardelyx design the trials and agreed to the clinical endpoints being chosen for the studies.
The FDA has asked the Company to conduct
“an additional adequate and well-controlled trial demonstrating a clinically relevant treatment effect on serum phosphorus or an effect on the clinical outcome thought to be caused by hyperphosphatemia in CKD patients on dialysis.”
This leaves the Company with a little chance for immediate or near-term approval. However, Tenapanor will enjoy patent protection until 2029 for composition, and to 2034 for use and may have a great chance of approval if it is able to demonstrate “PO4+ lowering or a head-to-head comparison w/ binder”, analysts at Jefferies wrote in a note to investors.
A Greater Chance for an Overseas Approval
In November 2017, Ardelyx entered into a licensing agreement with Kyowa Kirin for development and commercialization of Tenapanor for cardiorenal diseases and hyperphosphatemia in the Japanese Market. Furthermore, the Company has a licensing agreement with Shanghai Fosun Pharmaceutical Industrial Development Company Limited for development and commercialization of tenapanor for IBS-C and hyperphosphatemia related to chronic kidney disease in China. The Company has also entered into a licensing agreement with Knight Therapeutics, Inc. for commercial rights to tenapanor for the treatment of IBS-C and hyperphosphatemia in the Canadian market.
The Company’s unique approach is based on recreating and understanding unexploited ion transport mechanisms that are present in the gastrointestinal tract and kidney. Ardelyx is developing a pipeline of candidates that are orally active with minimal systemic absorption that reduce side-effects and drug-induced interactions. Ardelyx is developing RDX013 for the treatment of hyperkalemia that targets potassium secretion through the lumen of the gut and aims to lower the levels of serum potassium. The Company is also exploring other potential indications that could benefit from its unique approach.
An area with a large unmet need!
The National Institute of Diabetes and Digestive and Kidney Diseases defines chronic kidney disease (CKD) as the inability of the kidneys to filter blood due to factors such as diabetes, heart diseases, high blood pressure, Glomerulonephritis, Interstitial nephritis and Vesicoureteral among others. The disease involves the kidneys losing their functioning ability gradually over a period of time and leads to a build of harmful fluids, electrolytes and wastes in the body.
There are minimal or almost no signs of the disease in the initial stages and a patient may not realize that they are afflicted with the condition, until the damage is done. Some of the early symptoms include swelling of feet and ankles, nausea, loss of appetite, dry and itchy skin, uncontrolled hypertension to name a few. Treatment approaches aim to limit the progression of damage by addressing the underlying cause, however in spite of controlling the underlying cause the disease may progress to end-stage kidney failure, requiring dialysis or a kidney transplant.
Hyperphosphatemia, characterized by abnormally high serous phosphate concentration levels, typically greater than 4.5 mg/dL (> 1.46 mmol/L) is one of the side effects of reduced kidney function. The disorder affects more than 550,000 Americans with chronic kidney disease (CKD) on dialysis and can also lead to cardiorenal morbidity and mortality. The current treatment regimen consists of treatment with phosphate binders that are inefficient in reaching and maintaining desired phosphorus levels.
According to a report by BBC research, the global market for chronic kidney disease is likely to reach $95.0 billion by 2023, growing at a CAGR of 3.8 percent from $79.0 billion in 2018. A rising geriatric population, increasing lifestyle disorders such as diabetes and hypertension and growing research and development will drive the growth in the market.
A bumpy ride!
In 2015, the Company had terminated an agreement with AstraZeneca and bought back the worldwide development and commercialization rights for Tenapanor and other NH3 compounds. Ardelyx shelled out $15 million upfront along with other future contingent payments, in addition to 10 million in R&D costs. The Companies had initially entered into an agreement in 2012 for development and commercialization of its portfolio of NH3 inhibitors. AstraZeneca had reported higher-than-expected levels of diarrhea in patients undergoing a phase 2 study.
Ardelyx had entered into an option and license agreement with Sanofi for rights to its small molecule NaP2b inhibitors, also called RDX002, for the treatment of hyperphosphatemia in ESRD patients. However, the agreement was terminated by Sanofi in September 2015.
In September 2019, the Company received an approval for IBSRELA® (tenapanor), intended for the treatment of irritable bowel syndrome with constipation (IBS-C) in adults.
Conclusion
Ardelyx has the option of working with the FDA to resolve the issues raised against Tenapanor or challenge the non-approval in court, which may not be easy or desirable. Alternatively, Ardelyx could explore its potential as a combination therapy with other phosphate binders and push for an approval as a monotherapy later. The Company has the option of requesting for the opinion of an advisory committee, or resubmitting information from a phase IV trials to the FDA.
The candidate has generated a lot of interest in the nephrology community and has received positive response from doctors, who are seeking an improvement over existing treatment approaches.
The Company had total cash, cash equivalents and short-term investments of $171.8 million as of June 30, 2021 and has implemented a restructuring plan on receipt of the CRL, which is expected to decrease its annual cash compensation costs by approximately $17 million.
Ardelyx plans to request a Type A meeting with the FDA, which should provide more clarity on the way forward for the Company.
Disclosure: I/we have no positions in any stocks mentioned, and no plans to initiate any positions within the next 72 hours.
Click here to please visit our detailed disclosure
References
https://www.niddk.nih.gov/health-information/kidney-disease/chronic-kidney-disease-ckd
https://www.mayoclinic.org/diseases-conditions/chronic-kidney-disease/symptoms-causes/syc-20354521
https://ir.ardelyx.com/news-releases/news-release-details/ardelyx-regains-nap2b-program-sanofi





No Comments