
27 Apr Ocugen approaches the Finishing Line in the COVID Vaccine race, but is it a winner?
Covid-19 has bought many biotech companies into the limelight, all promising a possible solution to the pandemic that has disrupted lives worldwide. In February 2021, Ocugen, Inc. (NASDAQ: OCGN) shares catapulted after news of the Company signing a binding letter of intent (LOI) with Bharat Biotech became public. The agreement will see the companies co-develop Bharat Biotech’s COVAXIN for the U.S Market. The vaccine candidate has already obtained an emergency use authorization in India and has demonstrated efficacy in Phase 1 and 2 clinical trials in India.
On April 22, 2021, the Company announced that its Co-development partner, Bharat Biotech, released positive data from a phase III trial of COVAXIN. In the second interim analysis, the candidate demonstrated an efficacy of 78% and 100 % in mild/ moderate and severe COVID-19 including hospitalization respectively. The trial was conducted in over 25,800 patients in the age-group of 18-91 in India and included 2,433 people above the age of 60 and 4500 with co-morbidities.
Dr. Shankar Musunuri, Chairman of the Board, Chief Executive Officer, and Co-founder of Ocugen, commented,
“We continue to be excited by the compelling second interim results of Bharat Biotech’s Phase 3 clinical trial. We believe that COVAXIN can help change the course of this pandemic by preventing severe COVID-19 disease including hospitalizations by 100% as well as significantly limit the spread of asymptomatic COVID-19 infections based on efficacy shown to date. We are dedicated to being a part of the solution to save lives from COVID-19 by bringing COVAXIN to the U.S. market. Based on a traditional vaccine platform that has a long-established safety profile, we believe COVAXIN is an important tool to add to our national arsenal in ending the pandemic.”
The primary endpoint of the study is the first occurrence of PCR-confirmed symptomatic (mild, moderate, or severe) COVID-19 with onset at least 14 days after the second study vaccination in serologically negative (to SARS-CoV-2) adult participants at baseline. The secondary endpoint is based on accruing more than 87 symptomatic cases of COVID-19. However, due to the recent surge in cases in India, 127 symptomatic cases were recorded, resulting in a point estimate of vaccine efficacy of 78% (95%CI: 61-88) against mild, moderate, and severe COVID-19 disease. The trial will be continuing to its pre-planned conclusion.
Ocugen intends to file for an EUA with the FDA and will receive a specified minimum number of doses from Bharat Biotech upon approval. Ocugen will retain 45 percent of the profit from the sale of COVAXIN in the U.S, as per its co-development, supply and commercialization agreement with Bharat Biotech, and will be assisted in technology transfer for manufacturing for the U.S market by the Indian Pharma Company.
Ocugen Inc (NASDAQ: OCGN)
Market Cap: $1.47B; Current Share Price: 11.59 USD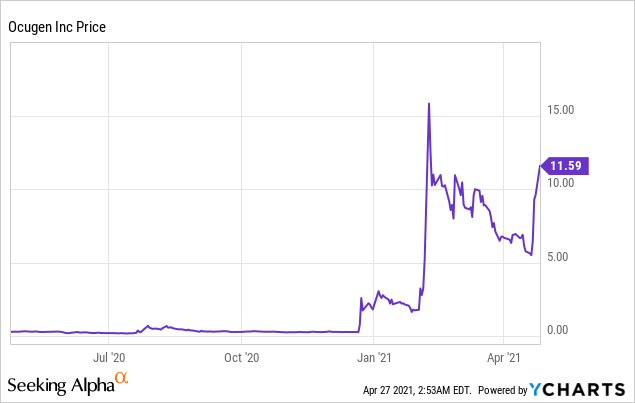
Data by YCharts
Industry
According to the World Health Organization (WHO), Coronavirus (CoV) is a family of viruses that cause illnesses ranging from common cold to lethal diseases such as the Middle East Respiratory Syndrome (MERS-CoV), Severe Acute Respiratory Syndrome (SARS-CoV) and a new strain of CoV named, novel coronavirus (nCoV). The virus is Zoonotic as it is transmitted from animals to humans, with the presence of many strains that have not yet infected humans. The infection manifests in the form of respiratory symptoms, cough, fever, shortness of breath and can take the form of pneumonia, severe acute respiratory syndrome, kidney failure and even death.
There is no definite cure for treatment or eradication of CoV, with treatment being limited to alleviation of symptoms, and providing support and care to patients. Scientists have been able to sequence the virus genetic code; spurring organizations around the world into action.
Company
Ocugen is a clinical-stage biopharmaceutical company that is focused on creating novel therapeutics to treat blindness diseases. The Company is using its modifier gene therapy platform to develop cutting-edge biologic product candidates for treatment of medical conditions with large unmet needs such as wet age-related macular degeneration, diabetic macular edema and diabetic retinopathy.

Image Source: Company
The Company’s pipeline consists of OCU400 intended for the treatment of Inherited Retinal Disorders. The candidate has received orphan drug designation in the U.S as well as EU, and has completed IND enabling studies so far. Ocugen is planning the initiation of Phase 1/2a in 2021. The other candidates in the pipeline include OCU410 for the treatment of Dry-Age Related Macular Degeneration and OCU200 for the treatment of Neovascular Disorders.
Furthermore, Ocugen has entered into a strategic manufacturing partnership with CanSinoBio (6185.HK) for OCU410 (AAV-RORA).
In June 2020, the Company discontinued the Phase 3 trial of OCU300, its lead candidate for ocular Graft vs. Host Disease (oGVHD).
Key Takeaways
Disclosure: I/we have no positions in any stocks mentioned, and no plans to initiate any positions within the next 72 hours.
Click here to please visit our detailed disclosure
References
https://ocugen.com/pipeline/overview/
https://ir.ocugen.com/static-files/f8198c96-a60e-4c55-a4c8-86b2a068f8f





No Comments