
20 May Onconova Upbeat about the prospects of its CDK 4/6 Inhibitors!
A Clinical-stage biopharmaceutical company, Onconova Therapeutics, Inc. (NASDAQ: ONTX) is developing novel therapeutics to combat cancer. The Company is developing a pipeline of small molecule candidates that target various forms of cancers such as Solid Tumors and KRAS+ NSCLC and a RAS pathway and a microtubule inhibitor intended for the treatment of COVID-19.
The Company’s approach combines a novel chemistry platform with cell-based differential screening. Onconova’s chemical library consists of over 150 core chemical structures and thousands of unique compounds consisting of two-ring or three-ring structures that are based on a signature element.
Unlike conventional Kinase Enzymes that require the binding of adenosine triphosphate, or ATP to be functional, Onconova’s library of molecules follow a more selective approach to inhibit kinase that does not require ATP binding.
Onconova’s ON 123300, is a proprietary multi-kinase inhibitor, which will start a dose-escalation and expansion phase 1 trial in the U.S in the second quarter of 2021. The candidate is already undergoing a dose-escalation and phase 1 trial in China. In addition, Oral rigosertib is being evaluated in a dose-escalation and expansion Phase 1 trial in patients with advanced KRAS+ lung adenocarcinoma in combination with nivolumab.
Rigosertib is also part of an investigator-initiated Phase 1/2 study, which is evaluating the drug in combination with nivolumab in KRAS mutated non-small cell lung cancer.
Onconova Therapeutics, Inc. (NASDAQ: ONTX)
Market Cap: $ 218.87M; Current Share Price: 0.925 USD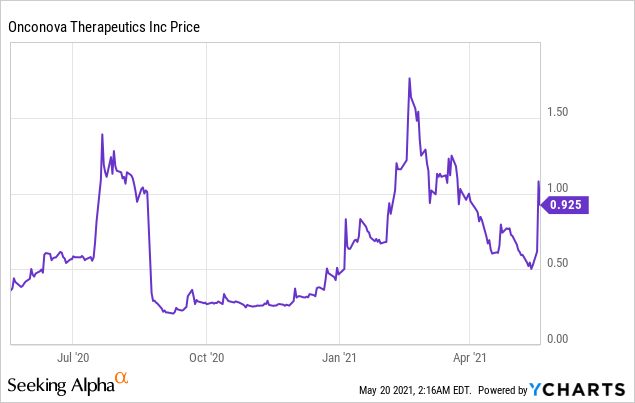
Data by YCharts
Commenting on the company’s performance in Q1, 2021, Steven M. Fruchtman, M.D., President and Chief Executive Officer of Onconova stated
“We are off to a strong start in 2021 and remain focused on advancing our clinical programs, in particular with our lead product candidate ON 123300, a multi-kinase inhibitor that potently targets CDK 4 and 6, which are overexpressed in a number of cancers, including HR+ HER 2- metastatic breast cancer, a potential blockbuster commercial opportunity.”
Industry
HER2 (human epidermal growth factor receptor 2), is a gene that plays a crucial role in the healthy functioning, growth and repair of breast cells. However, in about 25% of breast cancers, the HER2 gene multiplies uncontrollably. An HER2 – positive breast cancer is likely to grow and spread more rapidly than HER-2 Negative cancers.
Breast cancer is the most common form of cancer in women with an estimated one in eight women and one in 1,000 men being diagnosed with breast cancer during their lifetime. According to data made available by centers for disease control and prevention (CDC) over 237,000 cases of breast cancer are diagnosed in women and about 2,100 in men in the U.S alone. Approximately 41,000 women and 450 men in the U.S. succumb to this malady every year.
The breast cancer therapeutics industry is likely to reach USD 38.4 billion by 2025 growing at a CAGR of 10.7% according to a report by Grand View Research, Inc. The increasing spending on mergers and acquisition, strategic collaborations and the rise in research and development efforts to stay ahead of competitors will drive the growth in the market. Lifestyle changes such as rise in age of childbearing, high intake of oral contraceptive pills, reduced breast feeding, and undergoing estrogen modifying drug treatments are causing leading to increased incidences of breast cancer according to the report, while the onslaught of generics could hamper the market from reaching its full growth potential.
The treatment plan for Breast Cancer consists of Surgery such as mammaplasty, mastectomy, lumpectomy, Radiation therapy, chemotherapy and oral medications. These come with their own set of limitations and side-effects. However, new advancements in technology are helping companies to develop novel therapeutics with targeted delivery, improved efficacy, reduced side-effects and more positive patient outcomes.
Lung cancers are categorized into Non-Small Cell Lung Cancer, the most common form of cancer with an incidence rate of over 85% that manifests as Adenocarcinoma, Large Cell Carcinoma and Squamous Carcinoma. Small cell lung cancer also known as Oat Cell Cancer, which spreads rapidly makes up for around 10% to 15% of the cases and finally Lung carcinoid or neuroendocrine tumors that are less than 5% of the diagnosis and grow slowly and rarely spread.
Lung cancers constitute 13% of all new cancer diagnosis and are the second most common cause of cancer. According to an estimate by the American Cancer Society, there will be over 228,150 new cases of lung cancer (116,440 in men and 111,710 in women) diagnosed in 2019, with about 142,670 deaths resulting from it eventually. The death rate for Lung cancer alone is higher than that of colon, breast, and prostate cancers combined.
Image Source: Freepik
The Global Lung Cancer market is estimated to grow at a CAGR of 13.2%, during 2018-2022, to reach 36.93 billion by 2022 according to a report by Market Research Future. The rise in geriatric population, pollution levels, negative lifestyle conditions such as Active and passive smoking, increased healthcare expenditure and rapid scientific and technological advancements will drive the growth in the market.
The emergence of biomarkers such as epidermal growth factor receptor (EGFR), Anaplastic lymphoma kinase (ALK) and K-ras mutations (KRAS 1) have made the identification and management of lung cancer more accurate and manageable. These also help determine the optimal treatment plan for a patient, thereby avoiding the burden of undergoing treatments that may not be appropriate and the related side-effects.
Company
The Company has entered into strategic collaborations and partnerships with pharmaceutical and biotechnology companies to create a diverse pipeline of candidates that utilizes its proprietary chemistry platform. In December 2019, the Company entered into a distribution, licensing and supply agreement for commercialization of rigosertib with Specialized Therapeutics Asia, a biopharmaceutical company with presence in South East Asia, Australia and New Zealand.
Furthermore, the Company has also entered into a distribution, licensing and supply agreement with Knight Therapeutics Inc (GUD.TO) for exclusive rights to commercialize rigosertib in Canada. In August 2019, the Company entered into a partnership with Mission Bio to use its Tapestri Platform to investigate Onconova’s rigosertib through clinical trials. Onconova has also signed a License Agreement with Pint Pharma GmbH for commercialization of rigosertib in Latin America.

Image Source: Company
Onconova has also entered into a cooperative research and development agreement (CRADA) with the National Cancer Institute (NCI) to conduct research, pre-clinical laboratory evaluation and clinical trials of rigosertib in pediatric cancer associated RASopathies. The Company has also entered into a license and collaborative development agreement with HanX Biopharmaceuticals, Inc. for development and commercialization of ON 123300 a first-in-class dual inhibitor of CDK4/6 + ARK5 in China. The candidate is currently undergoing a dose-escalation and phase 1 trial in China.
Symbio Pharmaceuticals Ltd has a collaboration with Onconova for the development and commercialization of rigosertib in Japan and Korea. SymBio has been granted an exclusive license in exchange for an upfront payment and milestone payments, which will be payable on completion of certain milestones associated with the development of rigosertib, along with sales milestone payment and royalties on net sales.
Key Takeaways
Disclosure: I/we have no positions in any stocks mentioned, and no plans to initiate any positions within the next 72 hours.
Click here to please visit our detailed disclosure
References
https://www.onconova.com/pipeline/
https://investor.onconova.com/static-files/5fe03f21-cc14-4820-a7a3-20e888a62b2c
https://www.breastcancer.org/symptoms/diagnosis/her2
https://www.grandviewresearch.com/press-release/global-breast-cancer-drugs-market
https://www.fool.com/investing/2019/05/10/heres-why-puma-biotechnology-fell-as-much-as-407-t.aspx





No Comments