
22 Mar Robotic Exoskeletons and Stem Cells offer hope to Paraplegics
Paralysis is a widespread disability that affects 1.7 percent of Americans, with over 5,357,970 people living with some form of paralysis according a study conducted by the centers for disease control and prevention (CDC) in association with 14 leading universities and medical centers. A research study estimates that there are nearly 5.4 million people living with paralysis between the ages of 18 and 64 years.
Ischemic strokes are the leading cause of paralysis, followed by spinal cord injuries (SCI) and multiple sclerosis. A staggering 41.8% of paraplegics are unable to work. SCI’s alone cost an estimated $40.5 billion annually in terms of healthcare costs; about $158 billion is spent on home care and nursing home services combined, with friends or family members providing care that is valued at $306 billion annually.
The global spinal cord trauma market which was valued at $2.28 billion in 2017 is projected to reach $3.04 billion by 2025 with a CAGR of 3.7 percent according to a report by Persistent Market Research. The increasing incidences of spine related injuries owing to motor accident, workplace injuries, stroke and cancer related motor disability along with hyperextension of the spine and vertebral dislocation will spur the growth in the market. An increasing regulatory support to reduce SCI’s coupled with scientific advancements in the robotics operated exoskeleton market as well as stem cell research will help the market grow.
However, the economic burden of the disease and lack of insurance coverage is a major challenge for paraplegics, making it difficult for them to seek and adapt technological advancements in the field. This is set to change as some companies are revolutionizing the treatment options for paraplegics by their path breaking innovations ranging from improved exoskeleton support systems to stem cell and small molecule based treatment solutions. We take a look at some of these companies below:
GTX Medical (Private)
The company which was formed after a split from the Swiss federal institute of technology Lausanne (EPFL) is developing path breaking technologies in the area of spinal neurostimulation to treat people with debilitating spinal cord injuries (SCI). The company’s Targeted Epidural Spinal Stimulation (TESS), which was developed based on the work of Professor Grégoire Courtine, is a neuro-stimulation implant that combines real-time motion feedback and bodyweight support training tools to restore functioning and rehabilitate the individuals suffering from spinal cord injuries.

Image Source: Company
It has the distinction of being the only company in the world that has an implantable neural stimulation system that can restore the walking function in paraplegics. The company published a study of the efficacy of its stimulation protocols which prove that burst stimulation and spatiotemporal stimulation profiles mitigate the cancellation of proprioceptive information leading to better control over motor neuron activity by using Space-Time specific Epidural Electrical Stimulation (ST-EES). It is planning to initiate a study in H2 of 2019 to focus on incomplete SCI’s due to lesions in the cervical or thoracic segments of the spinal cord with individuals suffering from paraplegia.

Image Source: Company
GTX is currently developing the Go-2 Targeted Epidural Spinal Stimulation (TESS) system and is looking forward to obtaining a CE certification and FDA approval. The company is a part of the EUREKA funded research & development projects (DISPERSE, RESTORE and WALKAGAIN), a Euro 12.2 million project, that has three Dutch and Swiss-based companies working in tandem and building on each other’s innovations to provide solution to paraplegia.
The company’s Chief Scientific Officer, Prof. Grégoire Courtine, work was awarded No.1 breakthrough of the year award in 2017 by MIT. It is currently a privately held company funded by the likes of LSP, Wellingtonpartners, GIMV, inkef capital and Netherlands Enterprise Agency. It is also the recipient of grants and awards from institutions such as Welcome, Venture Leaders, Stimulus, PERL, Mass Challenge and the European Research Council.
Ekso Bionics Holdings (NASDAQ: EKSO)
Market Cap: $144.70M; Current Share Price: 2.31 USD
A world leader in the robotic based stroke and spinal cord injury rehabilitation exoskeletan market; it aims to use technology to improve human strength, endurance and mobility. With over 260+ rehabilitation centers in 30+ countries globally, EksoGT ensures correct movement patterns in recovery and assists in their progress towards independent ambulation. It offers various benefits with its proprietary technology such as a hand-held controller, real-time adjustable software, SmartAssist Software and EksoPulse Analytics.

Image Source: Company
EksoGT, its robotic skeleton is the world’s first commercially available solution for hemiplegia due to stroke and spinal cord injuries (SCI) approved by the FDA in the U.S and CE mark approved for lower-limb frailties in the European and Asian market. The company’s proprietary technology is protected by 30 U.S patents issued and over 200+ patents pending internationally. Its technology offers benefits such as improved endurance, functional independence, enhanced balance and midline alignment.
It also manufactures EksoVest for industrial use to minimize fatigue and risk of injury and has recently obtained a purchase order from two global aerospace manufacturers for use in assembly production lines of airplanes for commercial and defense use. The company has a tie-up with Ford (NYSE: F) for use of EksoVest on its automotive assembly line.
It also manufactures EksoZeroG that helps lessen the burden of construction and demolition workers.

Image Source: Company
As per its financial results for the full year 2018, its revenues stood at $11.3 million as compared to $7.4 million in 2017. It shipped a total of 90 EksoGT™ units in 2018 as against the 61 units shipped in 2017. Its cash and cash equivalents stood at $7.7 million.
Ekso Bionics entered into a $100 million joint venture with Zhejiang Youchuang Venture Capital Investment Co., Ltd (ZYVC) and Zhejiang Silicon Paradise Asset Management Group for creating a global manufacturing centre in Zhejiang and serving the exoskeleton market in China, Hong Kong, Singapore and Malaysia. As part of the agreement, it will license its manufacturing technology and patent rights in exchange for a 20% ownership position in consideration for loyalties based on mid-single digits percentage of the net sales revenue, while ZYVC and other partners will contribute over $90 million in cash and make a $10 million equity investment in Ekso.
Neuralstem (NYSE: CUR)
Market Cap: $8.58M; Current Share Price: 0.4713 USD
The companies NSI-566, a spinal-cord derived neural stem line, is its lead candidate for treatment of Amyotrophic Lateral Sclerosis (ALS), AIS-A complete chronic spinal cord injury (cSCI) and ischemic stroke related debility. Its proprietary technology facilitates the isolation of neural stems from the brain and spinal cord for generation of neurons that can replace diseased or dead cells.
The company has three proprietary technologies namely the small molecule program, the stem cell therapy program, and a new chemical entity screening platform. It has plans for an aggressive expansion of its pipeline of therapeutic products by evaluating new technologies that can create immense value.

Image Source: Company
Neural stem received an Orphan Drug Designation (ODD) for its NSI-566 for the potential treatment of ALS, a disease characterized by the death of neurons which disrupts communication to muscles and causes weakening of muscles, twitching and motor disability. NSI-566 has shown promise in clinical trials by protecting and nurturing these motor neurons. It has received positive results from both its phase I and Phase II clinical studies.
Image Source: Company
NSI-566 is also targeting Chronic Spinal Cord Injuries (cSCI), caused by either trauma, dislocation or the hyperextension of the cord related to motor accidents, workplace and sports related injuries as well as non-traumatic injuries related to cancer, intervertebral disc disease and vertebral injury to name a few. It has the potential to cover gaps in the spinal cord circuitry caused by traumatic SCI.
The company is currently engaged in Phase I trials in China through its wholly owned subsidiary, Suzhou Neuralstem, for use of NSI-566 in motor deficits caused by ischemic strokes. Paralysis is one of the most common after effects of ischemic stroke and can be permanent in nature. NSI-566 can establish a new circuitry and repair damaged cells to improve motor functions in stroke affected patients.
The company received an estimated $2.1 million in gross proceeds on account of an agreement with institutional investors for the purchase of 3,000,000 shares of its common stock, at a purchase price per share of $0.70, in a registered direct offering as per a company statement.
It’s cash, cash equivalents and investments as of September 30, 2018 stood at $5.7 million as per the company’s financial results for Q3, 2018.
Rewalk Robotics (NASDAQ: RWLK)
Market Cap: $23.97M; Current Share Price: 0.26 USD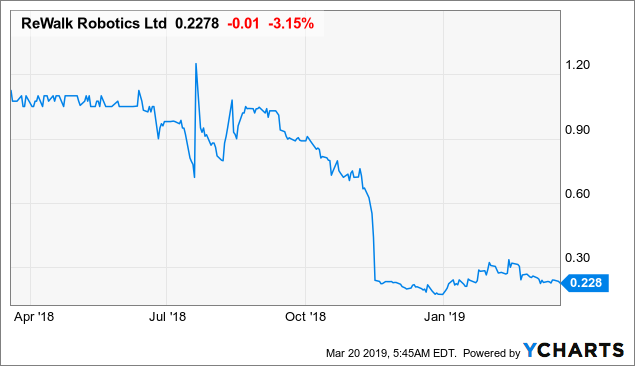
The company’s ReWalk robotic exoskeleton is the first one to receive FDA approval for personal and rehabilitation use in the United States. It also offers ReWalk rehabilitation for use in clinics for exercise and therapy. Its aim is to develop robotic technology that can improve the quality of life for paraplegics and people with lower limb disability.
It uses a patented motion sensing technology that is controlled by on-board computers and software and can be operated by individuals independently to walk, stand upright and turn.

Image Source: Company
ReWalk Personal 6.0 System is a customizable exoskeleton that offers safety along with optimal functioning. It can sense the motion of the body to generate steps and mimic a natural gait along with controlling changes in the centre of gravity. The company also offers comprehensive reimbursements, maintenance and warranty programs. It has training centres worldwide that can assist an individual in getting accustomed to the system.
ReWalk Rehabilitation is for clinical use for patients in need of therapy and exercise. It is customizable to suit individual patients and can be controlled by the therapist by means of a graphic user interface that allows for providing inputs on various parameters. The company offers a WW training standards program for the rehabilitation team to familiarize them with the technology.
The company has seen a positive response from numerous regulatory and insurance agencies, which have modified their policies to allow for the coverage of exoskeletal systems such as U.S department of Veteran Affairs which has expanded access to ReWalk for paralyzed veterans. It has also received coverage under the German Statutory Health Insurance Funds, Italian Ministry of Labor and Social Policy’s statutory insurance corporation (INAIL) to name a few.
The company recently celebrated a milestone of placing its 500th exoskeleton system in November 2018.
Asterias Biotherapeutics [acquired by BioTime (NYSE: BTX)]
Market Cap: $243.50M; Current Share Price: 1.57 USD
A biotechnology company that is creating novel solutions in the field of regenerative medicine, Asterias proprietary immunotherapy and pluripotent stem cell platform technologies are the basis of its innovations. It’s AST-OPC1 (oligodendrocyte progenitor cells) is currently in a Phase I & IIa clinical trials for application in SCI. Asterias has received a Regenerative Medicine Advanced Therapy (RMAT) designation for its AST-OPC1 from the FDA.

Image Source: Company
It’s lead candidate for SCI is AST-OPC1, that uses oligodendrocyte progenitor cells manufactured using its proprietary pluripotent stem cell platform. The company is currently enrolling patients affected by cervical (neck) spinal cord injury in Phase I & IIa clinical trials that are being funded by California Institute for Regenerative Medicine (CIRM). AST-OPC1 cells are surgically injected into the spinal cord which then nurture the healthy motor neurons and improve their transmission ability to restore mobility.

Image Source: Company
The company also has candidates under development for applications in oncology. Its AST-VAC1 is for the treatment of Acute Myeloid Leukemia (AML), while AST-VAC2 is an allogeneic (non-patient specific) immunotherapy for use in numerous cancers.
It has entered into two agreements with an affiliate of Novo Nordisk (NYSE: NVO) for a consideration of $2.0 million in upfront payment in September 2018.
Asterias reported cash and cash equivalents balance of $8.5 million and $6.2 in marketable equity securities as of October 1, 2018, as per its Q3, 2018 financial results; this includes the upfront payment from Novo Nordisk.
Source:
http://s3.amazonaws.com/reeve-assets-production/AJPH-Paralysis-Article-2.pdf
https://www.christopherreeve.org/living-with-paralysis/stats-about-paralysis
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5024361/
https://www.gtxmedical.com/about-us/
https://www.eurostars-eureka.eu/content/patient-broken-spine-walks-again
https://rewalk.com/category/press-releases/
Disclosure: I/we have no positions in any stocks mentioned, and no plans to initiate any positions within the next 72 hours.







No Comments