
19 Jul Will Aldeyra Hit the Bull’s Eye in Dry Eye?
Aldeyra Therapeutics, Inc. (NASDAQ: ALDX) announced positive results from the Phase 3 tranquility trial, which achieved its primary endpoint of being statistically superior to the vehicle for each of the two prespecified primary endpoints, Schirmer test (p=0.0001) and ≥10 mm Schirmer test responder proportions (p<0.0001) after a single day of dosing.
The Company intends to submit an NDA, pending discussions with the FDA, which will include ocular dryness symptom score, ocular redness, Schirmer test, and ≥10 mm Schirmer test responder analysis, along with results from five completed clinical trials. Aldeyra believes that the NDA will be the most comprehensive NDA submission in dry eye disease to date and has the potential to be the first to score an approval with symptoms and at least two labeled objective signs.
Aldeyra is scheduled to meet the FDA for a Type B Pre-NDA meeting in Q3,2022, with a potential NDA submission likely to follow. The Company is currently engaged in an ongoing 12-month safety trial pending enrollment and awaiting results from a dry eye chamber crossover trial due in Q3,2022. The results from the crossover trial will be submitted to the NDA as a supportive trial.
Aldeyra Therapeutics, Inc. (NASDAQ: ALDX)
Market Cap: $278.68M; Current Share Price: $4.78
Data by YCharts
Strength
Aldeyra’s lead product candidate is reproxalap, a small molecule RASP (Reactive Aldehyde Species) inhibitor for treating dry eye disease and allergic conjunctivitis. The Company is also developing ADX-2191 (Dihydrofolate Reductase) and ADX-629 for treatment of ethanol toxicity, chronic cough, Sjögren-Larsson Syndrome, and Minimal Change Disease. These three distinct biological mechanisms of action mentioned above have shown the potential to reduce levels of pathological inflammation through down-regulation of immune cell activation or proliferation.
The Company seeks to leverage its understanding of Reactive Aldehyde Species (RASP) levels to tackle Intracellular inflammatory factors, including NF-kB, a prominent protein in the inflammatory response. Its reproxalap can bind to free aldehydes, triggering covalent reproxalap-aldehyde adducts that can degrade intracellularly within hours, significantly lowering aldehyde levels. The candidate has undergone multiple Phase 2 and Phase 3 clinical trials in dry eye disease and allergic conjunctivitis and has shown statistically significant and clinically relevant improvements in ichthyosis (a severe skin disorder) caused by Sjögren-Larsson Syndrome.
Furthermore, the company has discovered additional RASP inhibitors, namely ADX-103 and ADX-629, which target retinal and autoimmune diseases. Aldeyra has partnered with Janssen to develop RASP inhibitors for systemic inflammatory diseases.
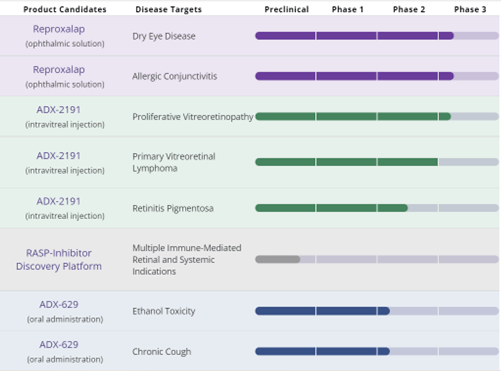
Image Source: Company
The company has acquired Helio Vision for its Phase 3-ready ADX-2191, an intravitreal DHFR inhibitor, for proliferative vitreoretinopathy (PVR). ADX-2191 has received Orphan Drug Designation from the U.S. Food and Drug Administration to prevent PVR. In addition, the Company in-licensed two clinical-stage candidates, namely ADX-1612 and ADX-1615 (an oral pro-drug of ADX-1612), developing them to treat various inflammatory diseases.
Weakness
In December 2021, the Company faced a setback with reproxalap failing to achieve the primary endpoint of ocular redness in the Tranquility trial, prompting Aldeyra to tinker with the other Phase 3 trial to obtain the desired result. Getting approval for Dry Eye requires data that demonstrates significant benefit on the signs and symptoms of the disorder in at least two clinical trials, as mandated by the FDA.
Aldeyra intends to submit data from part one of phase 3 Renew trial and results from the phase 2 Formulation study that demonstrates the candidate’s benefit on ocular dryness for the symptoms part of the stipulation. In the case of Signs, the Company will include data from a Phase 2 study that showed improvement in ocular redness with reproxalap versus vehicle.
However, the Tranquility trial achieved its secondary endpoint, the Schirmer test, which measures tear production. The Company has made ocular redness and Schirmer test as co-primaries, which means the trial will be a success if it achieves either of the endpoints. Aldeyra also increased the patient size from 300 to 400 for the Tranquility-2 trial.
Even if approved, the Company will face tough competition from the likes of Bausch (NYSE: BLCO) (NOV03), which plans to file for an NDA in the first of 2022 based on positive results from Gobi and Mojave, two pivotal trials.
Opportunity
Dry Eye Syndrome is characterized by the inability to produce a sufficient amount or quality of tears, which leads to a lack of lubrication and nourishment to the eye surface and manifests as chronic eye irritation, inflammation, and damage to the surface. Over 16 million diagnosed cases of dry eye syndrome in the U.S alone.
Dry Eye leads to discomfort and pain and can significantly impact the quality of life by making it difficult to perform everyday functions such as reading, driving, using computers, and completing daily activities. In the U.S, the financial implications of dry eye management were estimated to be US$ 11,302 per patient and US$ 55 billion overall, according to a report.
There is currently no cure for dry eye disease, and treatment options include topical medication, punctal plugs, practitioner visits, and nutritional supplements; these options are highly inadequate, hence the need for novel solutions. The prevalence of dry eye ranged from 5 to 30 % of individuals over 50, according to an estimate by The Epidemiology Subcommittee of the 2007 DEWS.
Fortune Business Insights reports that the industry will be worth USD 6.54 billion by 2027, growing at a CAGR of 4.7%. A rising geriatric population, increased screen exposure, diabetes, environmental factors such as pollution, and refractive surgeries are leading to a rise in the incidences of dry eyes. At the same time, the lack of good therapeutic options, expensive medication, and lack of patient adherence is a challenge to the growth of the market.
However, the market is poised for change with some novel technologies and drug candidates being developed to treat this chronic disorder. We take a look at some promising therapeutics in the offing.
Threats
Clinical Trials are fraught with risk and uncertainty. There is a possibility that the candidates in the Company’s developmental pipeline may not be able to meet their clinical endpoints in trials.
The Company may fail to receive regulatory approval for any other candidates, resulting in a setback for the other candidates in the pipeline.
However, a diverse pipeline will help mitigate the risk in case of adverse results or the failure to meet endpoints in any of its ongoing trials. The success of its clinical trials will allow the Company to advance its pipeline, but it should also be prepared to face any setbacks in case its ongoing attempts fail to meet its endpoints.
Key Takeaways
Disclosure: I/we have no positions in any stocks mentioned, and no plans to initiate any positions within the next 72 hours.
Click here to please visit our detailed disclosure
References
https://www.evaluate.com/vantage/articles/news/trial-results/aldeyra-needs-more-tranquility
https://ir.aldeyra.com/static-files/4f8e01a7-e242-404f-ba27-f3aac5699132





No Comments