
24 May 4 Reasons Why You Should Put Surface Oncology on your Watchlist!
A clinical stage Immuno-oncology company, Surface Oncology, Inc. (NASDAQ: SURF) is developing a diverse pipeline of candidates that target the immune system to combat various forms of cancer. The Company’s pipeline consists of candidates in four key areas namely macrophages and natural killer cells, T-Cell Inhibitors, blocking suppressive cell signalling molecules and cytokines and metabolites.
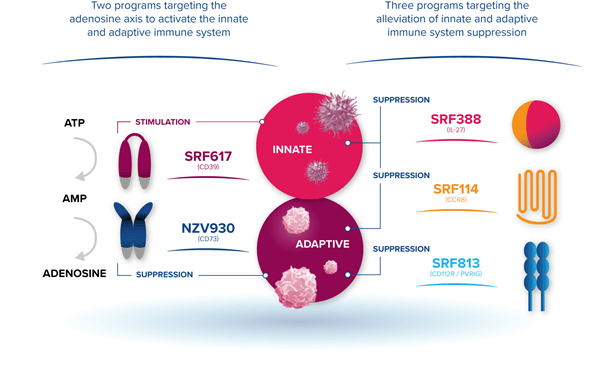
Image Source: Company
A Multi-Billion Dollar Industry
The Global market for therapeutic monoclonal antibodies (mAb) is expected to reach $114.6 billion by 2022, growing at a CAGR of 6.3% from $84.5 billion in 2017 according to a report by bbcresearch.com. Monoclonal Antibodies consist of identical immune cells derived from a single parent cell and have revolutionized the way therapeutics are developed. Monoclonal antibodies have the ability to bind specifically to cells and proteins and stimulate the immune system to fight foreign cells and possess characteristics such as affinity to antigens and antigen specificity.
The treatment market is divided into cancer, autoimmune diseases, hematological and dermatological disorders, with breakthrough treatments emerging in the field of cancer. There is a growing demand for human antibody therapeutics though there are other sources from which antigens can be derived such as chimeric, humanized etc.
The demand for mAb is driven by increasing research and development owing to government and regulatory impetus to the industry. Furthermore, the rising demand for personalized and customized therapeutics and the fewer side effects and specificity offered by mAb will contribute to the growth in the market.
Surface Oncology, Inc. (NASDAQ: SURF)
Market Cap: $ 378.70M; Current Share Price: 8.72 USD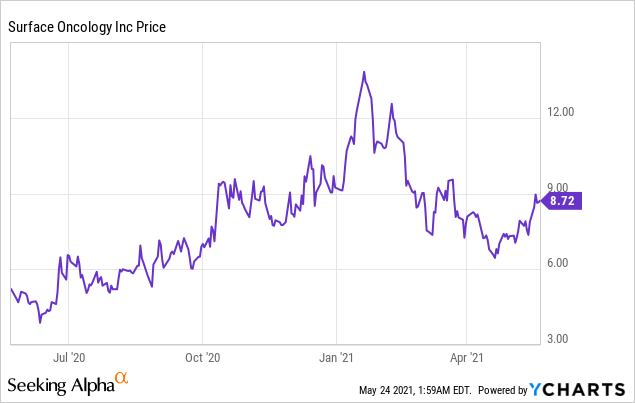
Data by YCharts
Pivotal Upcoming Catalysts
The U. S Food and Drug Administration granted an orphan drug designation for SRF617, intended for the treatment of pancreatic cancer in March, 2021. Furthermore, the Company will be sharing data from the Phase 1 trials of both SRF617 and SRF388 at the American Society of Clinical Oncology (ASCO) 2021 Annual Meeting, to be held virtually June 4-8, 2021.
A Promising Pipeline
Surface Oncology’s wholly-owned pipeline consists of the SRF617 program, a fully human antibody intended to target CD39, which plays a pivotal role in the production of adenosine and the breakdown of adenosine triphosphate (ATP). The candidate has demonstrated the potential to inhibit the enzymatic activity of CD39 in preclinical studies, both as a single agent and in combination with other therapies. The candidate is undergoing a phase 1/1b first-in-human dose escalation study in patients with solid advanced tumors. Additional studies are being planned with gemcitabine and abraxane; with Merck’s KEYTRUDA® (pembrolizumab), an anti-PD-1 antibody; or with AB928, an A2A/A2B small molecule inhibitor (in clinical collaboration with Arcus Biosciences).
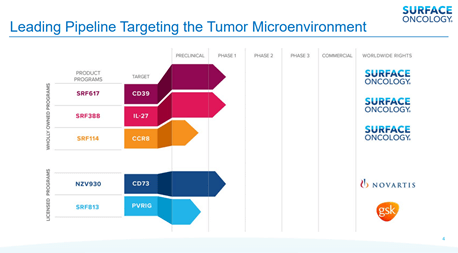
Image Source: Company
Furthermore, the Company is developing SRF388, a fully human anti-IL-27 antibody, which is currently being evaluated in a phase 1 first-in-human dose escalation study in patients with advanced solid tumors. In addition, SRF114 (CCR8) is also undergoing preclinical studies currently.
Multiple Strategic Collaborations
In March 2021, the Company entered into a clinical collaboration with Merck (NYSE: MRK), to evaluate the safety and efficacy of SRF388, an antibody therapy targeting IL-27, in combination with KEYTRUDA® (pembrolizumab), the first anti-PD-1 therapy approved in the United States. The study will be a part of the first-in-human Phase 1 study of SRF388, with a focus on patients with liver cancer and kidney cancer.
The Company has strategic clinical collaborations with Novartis and GSK to advance cancer therapies that address large unmet medical needs. As part of its collaboration with GlaxoSmithKline, the Company has granted exclusive worldwide licensing rights to SRF813, a fully human IgG1 antibody targeting PVRIG (CD112R), which acts as an inhibitory protein expressed on natural killer cells (NK cells) and T cells. The deal involves an upfront payment of $85 million along with an additional $730 million in future milestone payments. The Company is also eligible to receive tiered royalties on global net sales. Surface also intends to file for an Investigational new drug (IND) application for SRF813, which is being developed in partnership with gsk in 2021.
In 2016, Surface entered into an agreement with Novartis to grant worldwide license rights to NZV930 (formerly SRF373), a fully human CD73 antibody, in exchange for $70 million along with potential milestone payments up to $525 million, as well as tiered royalties.
Key Takeaways
Disclosure: I/we have no positions in any stocks mentioned, and no plans to initiate any positions within the next 72 hours.
Click here to please visit our detailed disclosure
References
https://www.grandviewresearch.com/press-release/global-monoclonal-antibodies-market
https://investors.surfaceoncology.com/static-files/8994cf65-5b18-41e5-a156-171c698f6954





No Comments