
14 Apr Rigel Pharmaceuticals has Multiple Aces Up its Sleeves!
Rigel Pharmaceuticals, Inc. (NASDAQ: RIGL) is focused on developing innovative small molecule drugs that target hematologic disorders, cancer and rare immune diseases. The Company announced positive topline results from a phase 2 evaluation of fostamatinib, an oral spleen tyrosine kinase (SYK) inhibitor, intended for the treatment of hospitalized patients with COVID-19. The candidate has met its primary endpoint and demonstrated a 50 percent reduction in Serious Adverse Events (SAEs). The trial is being conducted in collaboration with the National Heart, Lung, and Blood Institute (NHLBI), part of the National Institutes of Health (NIH), and Inova Health System.
In April 2021, the Company entered into a licensing agreement with Eli Lilly (NYSE: LLY) to co-develop and commercialize Rigel’s R552, a receptor-interacting serine/threonine-protein kinase 1 (RIPK1) inhibitor, indicated for the treatment of autoimmune and inflammatory diseases. Lilly will lead the clinical development of penetrating RIPK1 inhibitors in central nervous system (CNS) diseases as per a company statement. Rigel has received $125 million as upfront cash payment and is eligible to receive an additional $835 million in potential development, regulatory, and commercial milestone payments. Lilly will be responsible for the global commercialization of R552, while Rigel has the rights to the U.S market.
Rigel Pharmaceuticals, Inc. (NASDAQ: RIGL)
Market Cap: $614.14M; Current Share Price: 3.61 USD
Data by YCharts
Raul Rodriguez, Rigel’s president and CEO of Rigel, commented,
“We are very excited to form this strategic partnership with Lilly. This collaboration will provide significant resources and expertise to support a broad investigation in multiple disease indications with our RIPK1 inhibitors. With Lilly’s extensive knowledge in immune and CNS diseases, they are our ideal partner to ensure the clinical and commercial success of our RIPK1 inhibitor program.”
TAVALISSE – An FDA-Approved oral spleen tyrosine kinase (SYK) inhibitor for chronic immune thrombocytopenia (ITP)
The Company’s first FDA approved product is TAVALISSE®* (fostamatinib disodium hexahydrate), an oral spleen tyrosine kinase (SYK) inhibitor, intended for patients with chronic immune thrombocytopenia (ITP), who have had an insufficient response to a previous treatment. ITP is a rare autoimmune disorder in which the immune system attacks platelets in the blood. The drug has demonstrated increased platelet count within a short period from start of treatment and a lasting treatment benefit of up to 3 years and beyond.
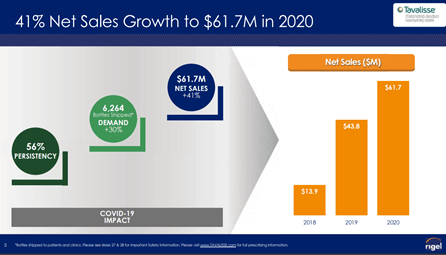
Image Source: Company
TAVALISSE® is also available in Europe under the brand name TAVLESSE and Canada as TAVALISSE. Fostamatinib is currently undergoing a Phase 3 clinical trial for the treatment of hospitalized COVID-19 patients, in addition to a NIH/NHLBI-sponsored Phase 2 clinical trial in hospitalized COVID-19 patients, in collaboration with Inova Health System. The candidate is also being evaluated in a Phase 2 clinical trial for the treatment of COVID-19 patients by the Imperial College London.
The candidate is currently under HC review in Israel and is undergoing a Phase 3 trial in Japan for the treatment of patients with chronic immune thrombocytopenia (ITP), who have had an insufficient response to a previous treatment.
Industry
Immune thrombocytopenia (ITP), formerly known as idiopathic thrombocytopenic purpura, is a rare autoimmune disorder that causes easy or excessive bruising and bleeding, caused by low levels of platelets. Symptoms generally include easy bruising, superficial bleeding or petechiae (usually in the lower legs), bleeding gums or nose, heavy menstrual flow among others. The condition may affect children after a viral infection and subsides without the need for any treatment. However, adults take a long time to recover and treatment includes medication to boost the platelet count or removal of spleen.
The condition may be caused by infections such as HIV, Hepatitis or H. pylori in adults and a viral infection such as mumps or flu in children. Young women are more vulnerable to this disease and risk is higher for those suffering from rheumatoid arthritis, lupus and antiphospholipid syndrome.
Treatment options include medication such as steroids, immune globulin, drugs to boost platelet production and in severe cases can lead to removal of spleen.
According to a report by Fortune Business Insights , the global immune thrombocytopenia market is poised to reach USD 3.33 Billion by 2026, growing at a CAGR of 0.54 percent, from USD 2.99 Billion in 2018.
According to the World Health Organization (WHO), Coronavirus (CoV) is a family of viruses that cause illnesses ranging from common cold to lethal diseases such as the Middle East Respiratory Syndrome (MERS-CoV), Severe Acute Respiratory Syndrome (SARS-CoV) and a new strain of CoV named, novel coronavirus (nCoV). The virus is Zoonotic as it is transmitted from animals to humans, with the presence of many strains that have not yet infected humans. The infection manifests in the form of respiratory symptoms, cough, fever, shortness of breath and can take the form of pneumonia, severe acute respiratory syndrome, kidney failure and even death.
There is no definite cure for treatment or eradication of CoV, with treatment being limited to alleviation of symptoms, and providing support and care to patients. However, Scientists have been able to sequence the virus genetic code; spurring organizations around the world into action to create drugs to fight the pandemic.
A Promising Pipeline
The Company’s pipeline consists of a phase 3 clinical evaluation of TAVALISSE in warm antibody autoimmune hemolytic anemia (AIHA), a blood disorder in which the immune system creates antibodies that damage haemolysis of the body’s own red blood cells (RBC). The candidate has already been granted an Orphan Drug designation and Fast Track designation by the FDA.
The Company’s lead candidate for the treatment of inflammatory conditions such as psoriasis, rheumatoid arthritis, lupus, multiple sclerosis, inflammatory bowel disease and gout is an investigational drug candidate named R835. The candidate is a potent and selective inhibitor of IRAK1 and IRAK4 that blocks inflammatory cytokine production in response to toll-like receptors (TLR) and the interleukin-1 family receptor (IL-1R) signalling as per a Company statement.
R835 inhibits the production of cytokine in response to TLR and IL-1R activation in vitro and is being tested in a phase 1 clinical study in healthy subjects. The study aims to prove the safety, tolerability and efficacy of R835 in inhibiting both the IRAK1 and IRAK4 signalling pathways, which play a crucial role in inflammation and immune responses to tissue damage.
Extensive Strategic Collaborations with Leading Pharmaceutical Companies
In June 2011, the Company entered into a strategic collaboration with BerGenBio for an exclusive worldwide research, development and commercialization agreement for R428 (now referred to as bemcentinib), a AXL receptor tyrosine kinase (AXL) inhibitor, indicated for the treatment of immune-evasive, therapy resistant cancers such as acute myeloid leukemia (AML), non-small cell lung cancer (NSCLC), triple negative breast cancer (TNBC), and melanoma. The Company has received an upfront payment and is eligible to receive milestone payments, sublicensing revenue as well as tiered royalties.

Image Source: Company
Furthermore, Rigel has entered into a collaboration with Daiichi-Sankyo (Daiichi) for development of Rain32 (DS-3032), an oral selective MDM2 inhibitor, indicated for the treatment of haematological malignancies such as relapsed/refractory AML and high-risk MDS. The Company has received an upfront payment and will receive milestone payments and royalties.
The Company is developing R256 (now referred as AZD0449), an inhaled Janus tyrosine kinase (JAK) inhibitor, in association with AstraZeneca (NASDAQ: AZN), for the treatment of chronic asthma. The candidate is currently being evaluated in a phase 1 clinical trial. Rigel has received an upfront payment and is eligible to receive developmental, regulatory, and launch milestones payments as well as tiered-royalties.
Rigel is also collaborating with Aclaris Therapeutics (NASDAQ: ACRS), Grifols, Kissei and Medison for the developing assets discovered in its laboratory.
Key Takeaways
Disclosure: I/we have no positions in any stocks mentioned, and no plans to initiate any positions within the next 72 hours.
Click here to please visit our detailed disclosure
References
https://www.rigel.com/pipeline
https://www.fortunebusinessinsights.com/industry-reports/immune-thrombocytopenia-itp-market-101682





No Comments