
28 Apr 5 Biotech Companies Likely to face Massive Hurdles Ahead!
Biotech Companies continue to show resurgence even in these challenging times, with more and more companies persevering with bringing their clinical candidates to fruition. While Vaccine development and therapeutics to combat the COVID-19 pandemic are at the forefront of development activities in the biotech sector, there are some companies that are making strides in other related fields such as oncology, NASH, HBV, or Chronic Kidney Disease. However, some of these companies have faced massive challenges such as failures of pivotal trials and FDA feedback that has put a spoke in wheel of their development plans. We take a look at some of them below:
Emergent BioSolutions (NYSE: EBS)
Market Cap: $ 3.42B; Current Share Price: 63.75 USD
Data by YCharts
Emergent BioSolutions, a life sciences company that was roped in to produce COVID-19 vaccines for both AstraZeneca and Johnson & Johnson has had a rough month so far. On April 19, 2021, the Company made a filing with the United States Securities and Exchange Commission that it has halted production of any new material at its Bayview facility at the request of the FDA, which had initiated an inspection of the facility on April 12, 2021. The Company would be required to quarantine all existing material that was produced at the facility, pending completion of the inspection and declaration of the findings.
The Company’s woes began in March, when the news of workers at the Bayview facility mixing up the ingredients and ruining a batch of nearly 15 million vaccine doses of J&J became public. Fortunately, the mistake occurred during the early production stages and none of the doses even made it to distribution. This resulted in a temporary halt in production of Johnson & Johnson’s vaccine at the facility, with the Company issuing a statement that it was stepping up to implement tighter controls and will ensure stringent quality checks and test runs to adhere to its high-quality standards.
Johnson and Johnson took over the responsibility of the manufacturing facility, while AstraZeneca moved the production of its viral vector product to another location.
Now the Company has received a damning FORM 483 from the FDA, which lists unsanitary conditions, inadequate facility size, substandard employee training and inadequate written procedures as some of the issues with its Bayview facility. Emergent has issued a press release stating that it is working closely with the FDA and Johnson & Johnson to address the issues as quickly as possible. However, the Company may find it difficult to land more manufacturing contracts in the near-term due to these events.
The Company’s product line consists of a smallpox vaccine namely ACAM2000 vaccine; ANTHRASIL, an anthrax immune globulin Intravenous; BAT, Botulism Antitoxin Heptavalent and Biothrax an anthrax vaccine adsorbed among others. Emergent has an extensive pipeline of candidates that target treatment of conditions such as Vaxchora Pediatric (Cholera Vaccine), AV7909 (Anthrax), CHIKV VLP (Chikungunya virus VLP vaccine), FLU – IGIV (seasonal influenza A virus therapeutic) to name a few.
In addition, the Company also has multiple platforms such as The Emergard® auto-injector platform intended for intramuscular rapid delivery as part of emergency response medical treatments; hyperimmune specialty plasma product manufacturing platform, broad-spectrum antiviral iminosugars platform and broad-spectrum antibiotics that target quinolone-resistant bacterial strains
Emergent has 8 manufacturing facilities that produce over 20 commercial products and has supported the development of more than 200 clinical candidates. In June 2012, the Company had entered into a public-private partnership with Biomedical Advanced Research and Development Authority (BARDA) to establish a Center for Innovation in Advanced Development and Manufacturing (CIADM). The Company’ s CIADM has already been awarded four task orders by BARDA to develop Ebola and Marburg therapeutics and a Zika vaccine.
Wave Life Sciences Ltd (NASDAQ: WVE)
Market Cap: $ 302.80M; Current Share Price: 6.18 USD
Data by YCharts
In March 2021, Wave Life Sciences announced the shelving of two of its most advanced drug candidates namely WVE-120101 and WVE-120102, based on early clinical testing data from the Phase 1b/2a PRECISION-HD2 and PRECISION-HD1 trials, which showed no statistically significant effect on the mutated proteins that cause Huntington’s disease.
In December 2019, the Company announced the decision to discontinue the development of suvodirsen for patients with Duchenne muscular dystrophy (DMD), who have mutations amenable to exon 51 skipping, based on the interim results from a phase 1 open-label extension (OLE) study. However, the Company plans to carry out the site activation and enrolment in the Phase 1b/2a clinical trial of WVE-003 intended for the treatment of Huntington Disease in adults that carry SNP3.
Furthermore, the Company will initiate dosing in two other clinical trials namely WVE-004 (targeting C9orf72) for amyotrophic lateral sclerosis (ALS) and frontotemporal dementia (FTD) and WVE-N531 (targeting exon 53) for Duchenne muscular dystrophy (DMD). Wave has already submitted a clinical trial application (CTA) for WVE-N531 in March 2021.
Wave Life Sciences uses the oligonucleotide approach to creating nucleic acid based therapeutics, for the treatment of rare genetic diseases, such as Huntington’s disease, amyotrophic lateral sclerosis and frontotemporal dementia. The Company’s proprietary platform can chemically modify short-length RNA or DNA strands to alter the production of disease-causing proteins and inhibit degradation of oligonucleotides by changing their natural phosphate backbone to phosphorothioates, by replacing a non-bridging oxygen atom with a sulphur atom. Its chemistry platform allows for the design, manufacturing and optimization of stereopure oligonucleotides.

Image Source: Company
The technology enables the company to create medicines with improved safety profiles, stability and far greater pharmacological properties, which have a wide applicability across platforms such as Antisense, Exon Skipping, Splice Correction, RNAi and MicroRNA. The company has a scalable in-house cGMP manufacturing unit in Lexington, Massachusetts, which helps it regulate the development and supply of its products.
The development of the company’s lead candidate for DMD suvodirsen (WVE-210201), which used exon skipping to create truncated functional dystrophin protein to target restoration of the mRNA reading frame, was discontinued in December 2019. The Drugs Phase 2/3 efficacy and safety trial for Duchenne muscular dystrophy (DMD) was selected for the complex innovative trial designs (CID) by the U.S. Food and Drug Administration (FDA) pilot program.

Image Source: Company
Wave has a global 50:50 co-development and co-commercialization profit share collaboration with Takeda Pharmaceuticals for developing therapies for the treatment of central nervous system (CNS) disorders. It has partnered with Pfizer for developing therapeutics for debilitating metabolic diseases by combining its stereopure platform, across antisense and RNAi modalities, with GalNAc and Pfizer’s hepatic targeting technology. In addition, it has a tie-up with ReadCoor for developing stereochemistry in optimizing silencing and splicing therapies in the brain.
It has multiple research collaborations such as with the University of Oxford for the study of its proprietary isomers to improve the pharmacology of oligonucleotides for the treatment of DMD and with University of Dundee for developing treatment for common and rare skin diseases using antisense oligonucleotides.
Wave Life Sciences has faced two setbacks in a row, the failure of its DMD candidate and the underwhelming results of its HD candidate, raising questions about the efficacy of its technology and the success of other candidates in the pipeline.
The HD clinical pipeline has nearly 23 candidates under various stages of clinical development, with the Company facing stiff competition from Ionis, which has an ongoing phase II trial in collaboration with Roche, the results from which are expected to be announced in late 2020. However, the approach that the Company is adopting is different from that of Roche, and if it succeeds in its mission, then it can wrestle market share from Roche even if the company manages to capture the first mover advantage.
The Company has robust strategic partnerships with Roche and Pfizer, which extends to other promising candidates in its pipeline such as ALS, FTD, Spinocerebellar ataxia 3, CNS Diseases with Takeda and Metabolic liver disease with Pfizer. This would help the Company mitigate the potential impact on its pipeline, while leveraging on the experience and support of these established players.
Inovio Pharmaceuticals Inc (NASDAQ: INO)
Market Cap: $ 1.50B; Current Share Price: 7.17 USD
Data by YCharts
The Company’s shares took a hit as news of the U.S government deciding to withdraw funding for its late-phase Innovate trial for INO-4800 intended for the treatment of COVID-19 vaccine became public. The decision will not affect the funding of its Phase 2 trial and will have no bearing on its other programs with the U.S government. The Company will continue the ex-US development of the vaccine candidate in association with its global partners such as Advaccine Biopharmaceuticals (China) and will be evaluating INO-4802, a pan-COVID vaccine candidate that could work against the current as well as the future strains of the disease.
Inovio Pharmaceuticals is focused on developing DNA-based immunotherapy and monoclonal therapies to fight cancer and infectious diseases. The Company’s immunotherapy enables a patient’s immune system to produce functional antibodies and killer T cells that are capable of combating cancer and infectious diseases. The technology helps identify strains and variants of a target cancer or virus, assess gene sequence of selected antigens, create an optimal consensus sequence for the antigen and insert a synthetic consensus sequence for each antigen into a separate DNA plasmid to manufacture SynCon Immunotherapy. The therapy can then be delivered into the muscle or skin using its Cellectra Device which causes the immune system to produce protective antibodies and killer T Cells to fight the infectious cells or cancer.
The Company’s proprietary CELLECTRA allows plasmid construction to be delivered via intramuscular or intradermal. It is an advanced transfection delivery that helps increase immunotherapy cellular intake and promotes antigen expression leading to CD8+ T cell activation and antibody response. Inovio has a diverse pipeline of candidates such as VGX-3100 meant for the treatment of Cervical Dysplasia, Vulvar Dysplasia and Anal Dysplasia (In collaboration with ApolloBio (China Exclusivity)). Furthermore, the Company is developing INO-3107 for the treatment of Recurrent Respiratory Papillomatosis (RRP), INO-5401 for the treatment of Glioblastoma, INO-5151 for Prostate Cancer (In association with Regeneron and Parker Institute for Cancer Immunotherapy)

Image Source: Company
Invoio is collaborating with AstraZeneca to for the development and commercialization of MEDI0457, a PD-L1 checkpoint inhibitor to be used in combination with durvalumab, intended for the treatment of metastatic HPV-related SCCHN. The Company is actively seeking strategic partnership and collaborations to fund and develop its platform of infectious diseases. In addition, the Company had entered into agreement with QIAGEN, to co-develop liquid biopsy-based pretreatment test to guide patient selection for VGX-3100, a potential first-in-class treatment for HPV Infection of the Cervix and the first non-invasive treatment for cervical pre-cancer.
Additionally, its partner funded development pipeline has various candidates such as PENNVAX-GP, a HIV therapeutic being developed in association with NAID. The company intends to release the topline efficacy and tolerability data from the Phase 3 REVEAL trials in VGX-3100 in H1,2021. Furthermore, Inovio is also planning to initiate a Phase 3 trials for VIN/AIN and will be targeting an orphan drug designation. The Company will also initiate a phase 2 study for Lassa with INO-4500 and MERS study with INO-4700, both being funded by CEPI.
Protara Therapeutics Inc (NASDAQ: TARA)
Market Cap: $ 162.80M; Current Share Price: 11.83 USD
Data by YCharts
Protara Therapeutics announced its plans to complete confirmatory, large-scale, GMP manufacturing comparability to a similar treatment in H2, 2021 as per feedback received from the FDA and will subsequently initiate a clinical study in pediatric of Lymphatic malformations (LM) patients subject to alignment with FDA on study design. The Company plans to continue development of TARA-002, derived from the same cell bank as OK-432, and has been granted a Rare Pediatric Disease designation by the FDA for the treatment of LMs.
OK-432 has already been approved in Japan and Taiwan for the treatment of Lymphatic malformations (LMs) and had successfully demonstrated initial manufacturing comparability between TARA-002 and the originator compound OK-432, in one of the largest Phase 2 trials ever conducted in LMs with more than 500 patients.

Image Source: Company
In addition to TARA-002, the Company is also developing an investigational phospholipid substrate replacement therapy for intestinal failure-associated liver disease (IFALD). The candidate has completed the end of phase 2 dialogue with the FDA. Protara intends to complete nonclinical studies and file an IND in H2,2021 followed by the initiation of a phase 1 study.
FibroGen, Inc (NASDAQ: FGEN)
Market Cap: $ 1.98B; Current Share Price: 21.53 USD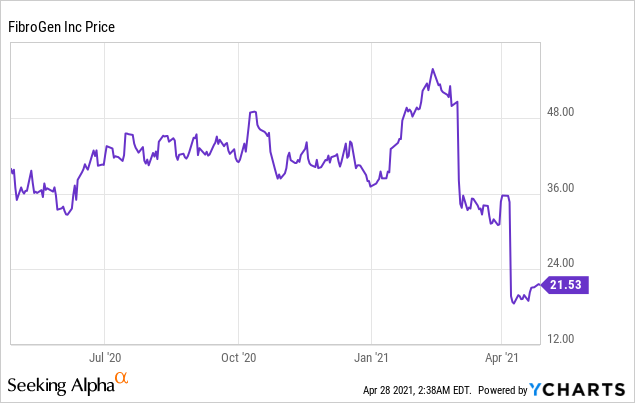
Data by YCharts
FibroGen is a biopharmaceutical Company leveraging its expertise in hypoxia-inducible factor (HIF) and connective tissue growth factor (CTGF) biology to create a diverse pipeline of first-in-class therapeutics. The Company has had a rough April so far, with shares of the company crashing on the heels of the disclosure that the safety analyses from a late-stage study of roxadustat in treating anemia of chronic kidney disease (CKD) included post-hoc changes to the stratification factors. The information essentially means that it can no longer claim that it is better at reducing major adverse cardiovascular events (MACE) when compared to erythropoiesis-stimulating agent epoetin alfa. The declaration comes at a time, when FibroGen is preparing to meet the U.S. Food and Drug Administration (FDA) advisory committee to discuss and review the regulatory filing for roxadustat.
Roxadustat, the Company’s lead candidate is a first-in-class, oral small molecule intended for the treatment of anemia associated with chronic kidney disease (CKD), which is currently being evaluated in phase 3 clinical trials. The hypoxia-inducible factor prolyl hydroxylase (HIF-PH) inhibitor has demonstrated the ability to improve iron availability and reduce hepcidin. The product candidate has already been approved in China, Japan, and Chile for the treatment of anemia in CKD patients on dialysis and patients not on dialysis.

Image Source: Company
Astellas, its development partner, had filed the Marketing Authorization Application for roxadustat in Europe, which was accepted by the European Medicines Agency for review in May 2020. The candidate is currently undergoing Phase 3 clinical development in the U.S. and Europe. In addition, it is also being evaluated in a Phase 2/3 development in China for anemia associated with myelodysplastic syndromes (MDS) and a Phase 2 U.S. trial for treatment of chemotherapy-induced anemia (CIA).
Furthermore, the Company is also developing Pamrevlumab, a fully-human monoclonal antibody, which is currently being evaluated in phase 3 clinical trials for the treatment of locally advanced unresectable pancreatic cancer (LAPC), Duchenne muscular dystrophy (DMD), and idiopathic pulmonary fibrosis (IPF). The candidate has been granted Orphan Drug Designation for LAPC, DMD, and IPF and Fast Track designation for LAPC and IPF. The candidate was recently awarded Rare Pediatric Disease Designation by the FDA for the treatment of Duchenne muscular dystrophy (DMD). The candidate is currently being evaluated in two Phase 3 trials in DMD.
The Company has strategic collaboration with Astellas for development of hypoxia-inducible factor (HIF) anemia programs since 2004 for Japan, Europe, the Middle East, the Commonwealth of Independent States, and South Africa. FibroGen has entered into a partnership with AstraZeneca to develop the anemia programs in the U.S and other markets not licensed to Astellas, including China.
Disclosure: I/we have no positions in any stocks mentioned, and no plans to initiate any positions within the next 72 hours.
Click here to please visit our detailed disclosure
References
https://investors.emergentbiosolutions.com/static-files/eeb6d4b8-ff62-41a5-9cdc-ed7b1214d576
https://www.pharmtech.com/view/j-j-to-take-over-its-product-manufacturing-at-emergent-facility
https://www.emergentbiosolutions.com/manufacturing-expertise
https://ir.wavelifesciences.com/static-files/d720add1-04ae-4206-bcaa-cb61f0e5a031
https://ir.protaratx.com/static-files/aed03220-d91c-4f24-a6b3-e0a51db8fd65





No Comments