
11 Oct Pieris’s Anticalin could be a Gamechanger!
Pieris Pharmaceuticals, Inc. (NASDAQ: PIRS) develops Anticalin-based drugs for treatment of conditions related to immuno-oncology and respiratory diseases such as Asthma and Anaemia. Anticalin proteins are engineered versions of lipocalins that are human proteins having the ability to bind, store and transport molecules, which the Company has leveraged for delivering drug candidates binding to a broad spectrum of therapeutic targets, in the clinic and in partnerships with leading pharmaceutical companies. The technology was licensed from the Technische Universität München (TUM) and provides it exclusive access to Anticalin-related IP.
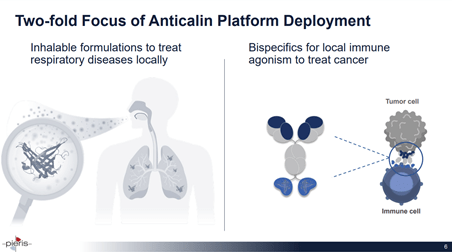
Image Source: Company
Anticalin Proteins are a novel therapeutic class that are derived from lipocalins and are smaller than mAbs (~18 kDa vs ~150kDa mAbs). They are suitable for inhalable delivery and can address unmet areas in respiratory disorders.
Pieris Pharmaceuticals, Inc. (NASDAQ: PIRS)
Market Cap: $316.88M; Current Share Price: 4.75 USD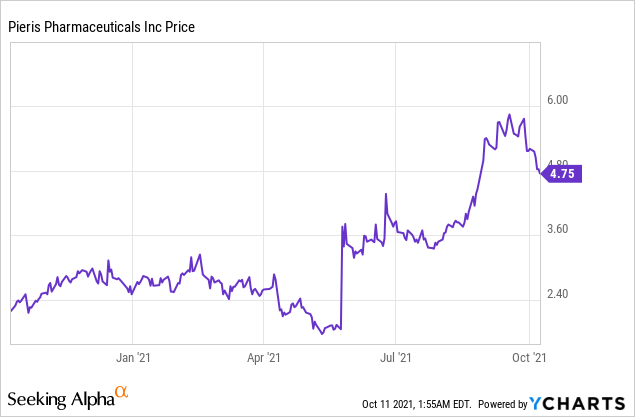
Data by YCharts
Strengths
Pieris Pharmaceuticals recently entered into a multi-program research collaboration and license agreement with Genentech, to develop and commercialize respiratory and ophthalmology therapies, using Pieris’ proprietary Anticalin® technology. The agreement involves an upfront payment of $20 million to Pieris, along with eligibility to receive another $1.4 billion in additional milestone payments as well as tiered royalties for any program that reaches commercialization. Pieris is responsible for the discovery, research and pre-clinical development of any potential candidate, while Genentech will handle IND-enabling activities, clinical development, and commercialization. Genentech can choose additional targets for an option exercise fee.
The Company is developing PRS-060/AZD1402, an inhaled IL-4 receptor alpha inhibitor, intended for the treatment of moderate-to-severe asthma in collaboration with AstraZeneca (NASDAQ: AZN). The candidate has initiated dosing in the first part of a global phase 2a study and expects to announce the result from the trial next year. In addition, Pieris is planning a two-arm phase 2 study for cinrebafusp alfa, a 4-1BB/HER2 bispecific for the treatment of HER2-expressing solid tumors, in gastric cancer, in collaboration with Lilly (NYSE: LLY) and Seagen (NASDAQ: SGEN). The first arm of the study will evaluate the combination of ramucirumab and paclitaxel in HER2-high gastric cancer, while the second arm will study the combination with tucatinib in HER2-low gastric cancer.

Image Source: Company
In 2017, the Company signed deals with Servier in immuno-oncology and is planning to present preclinical data for PRS-344/S095012, a 4-1BB/PD-L1 bispecific, at the AACR Annual Meeting 2021. The candidate will advance into phase 1 development this year. Pieris holds the exclusive commercialization rights in the U.S and is eligible for royalties on ex-us sales. Both companies are also developing PRS-352, an undisclosed bispecific. Pieris has a strong patent portfolio, comprising key patents granted in the United States, Europe and Asia, covering the Anticalin® protein as a drug class, libraries of Anticalin proteins and all derived products.
Weakness
The Company does not yet have a successful commercial product in the market. The Company has a promising pipeline and strategic partnerships with leading biopharmaceutical companies, however none of these candidates are in phase 3 trials and are years away from gaining an FDA approval.
Opportunity
Asthma is a respiratory condition in which the airways narrow, swell or produce excess mucus leading to breathing difficulty, wheezing or coughing. The disease can severely impact day-to-day life and may even result in a life-threatening attack. Most of the common symptoms include shortness of breath, tightness or pain in the chest, wheezing and trouble sleeping. The disorder can progressively worsen over time and will manifest in the form of more frequent asthma attacks, poor readings on a peak flow meter or increasing usage of inhalers to ease the discomfort.
The condition can be triggered by certain situations such as physical exertion or exercise, change in weather, exposure to chemicals or fumes at the workplace, or even exposure to allergy-causing substances such as pollen, molds, or pet dander.
Though the disease has no cure at present, it can be managed through use of medication. The condition needs to be monitored carefully to prevent extensive long-term lung damage and effective management of the condition, with minimum disruption to daily life. Risk factors include family history, preexisting conditions such as atopic dermatitis, obesity or constant exposure to second-hand smoke.
According to a report by Precedence Research, the global asthma drugs market is expected to reach USD 37.3 billion by 2030, growing at a CAGR of 5.2 percent from USD 20.6 billion in 2020. The rise in pollution levels, increasing cigarette smoking, growing research and development investments by pharmaceutical companies are likely to drive the growth in the market.
Threats
Clinical Trials are fraught with risk and uncertainty. There is a possibility that the candidates in the Company’s developmental pipeline may not be able to meet their clinical endpoints in trials. However, a diverse pipeline will help mitigate the risk in case of adverse results or the failure to meet endpoints in any of its ongoing trials. The success of its clinical trials will help the Company advance its pipeline but it should also be prepared to face any setbacks, in case its ongoing trials fail to meet their endpoints.
Key Takeaways
Disclosure: I/we have no positions in any stocks mentioned, and no plans to initiate any positions within the next 72 hours.
Click here to please visit our detailed disclosure
References
https://www.mayoclinic.org/diseases-conditions/asthma/symptoms-causes/syc-20369653
https://finance.yahoo.com/news/pieris-pharmaceuticals-receives-orphan-drug-120000808.html





No Comments