
03 May Will Evelo Biosciences Differentiated SINTAX approach pay off?
Evelo Biosciences, Inc. (NASDAQ: EVLO) is focused on developing innovative oral biologics that target cells in the intestines for the treatment of a range of inflammatory diseases and cancer. The Company’s approach is to develop systemically acting biologics that can act on the cells in the small intestine or more specifically the small intestinal axis, SINTAX™, which plays a critical role in regulating key immune, metabolic and neurological systems.
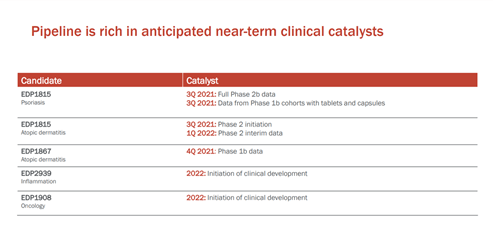
Image Source: Company
The Company has some key upcoming catalysts such as the full data from Phase 2b dose-ranging trial of its lead candidate EDP1815 in psoriasis, the initiation of a Phase 2 trial in atopic dermatitis in Q3, 2021, the Interim safety and futility analysis from Phase 2/3 TACTIC-E trial in Q2, 2021, which is studying the safety and efficacy of EDP1815 for treatment of newly hospitalized patients with COVID-19 and initiation of clinical development of EDP2939 and EDP1908 in Inflammation and Oncology respectively.
Evelo Biosciences (NASDAQ: EVLO)
Market Cap: $702.14M; Current Share Price: 13.15 USD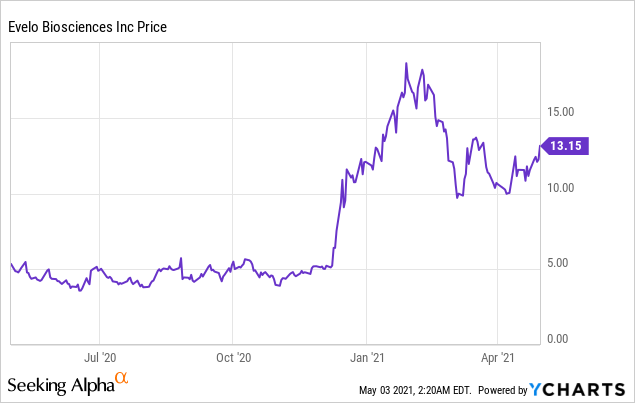
Data by YCharts
Industry
Atopic Dermatitis, also known as Eczema, is a skin condition caused by a gene variation and is characterized by red and itchy skin. The disease commonly occurs in childhood but may present at any age. The condition is chronic in nature and flares up from time to time and is usually accompanied by asthma and hay fever. The disease manifests in the form of dry skin, uncontrollable itching, rash or patches on arms, hands, feet and in the crevices of the elbows and knees to name a few. In addition, there may be raised bumps on the skin, which may leak fluid and cause the skin to harden and become cracked and scaly.
Though there is no cure for Atopic Dermatitis at present, the condition can be managed by using ointments and lotions to seal in the skin’s moisture, avoiding triggers that can worsen the condition such as sweating, soaps, detergents or pollution and using a beach bath as recommended by the American Academy of Dermatology.
According to a report by Market Data Forecast, the global atopic dermatitis drugs market is projected to reach USD 19.3 billion by 2025, growing at a CAGR of 13.12 percent from USD 10.4 billion in 2020. The growing incidences of seasonal allergies, climatic changes and pollution are driving the growth in the market.
Company
The Company is keen on exploring the unrealized potential of SINTAX and creating transformative therapies that have the potential to change the treatment landscape in inflammatory diseases and cancer. Microbes play a crucial role in conditioning the immune cells in the small intestine and hence the Company is working on developing orally delivered, single strains of microbes selected for defined pharmacological properties. Evelo believes that specific microbes can downregulate or upregulate immune responses through their effect on SINTAX, and therefore its approach involves engaging with the host immune cells during their journey through the small intestine.
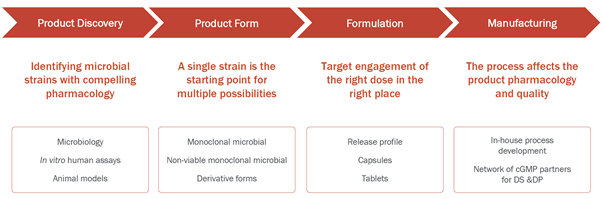
Image Source: Company
Evelo has a robust intellectual property rights portfolio with multiple issued patents and pending applications that protect its product candidates and platform technology. The Company’s pipeline consists of EDP1815 and EDP1867 that are currently undergoing clinical development for the treatment of inflammatory diseases. EDP1815, its lead product candidate is being evaluated in multiple clinical trials such as a phase 2b dose-ranging trial in psoriasis, the full data from which is expected to be available by Q3,2021.
EDP1815 is also being evaluated in a phase 2 Rutgers University trial, in a double-blind placebo-controlled setting, to evaluate the safety and efficacy of EDP1815 for hospitalized patients with newly diagnosed COVID-19. The candidate will be tested for its potential to prevent the progression of the disease and development of COVID-Related Complications (CRC).
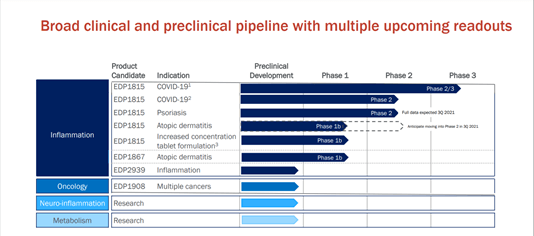
Image Source: Company
In addition, the candidate is also being studied in a Phase 2/3 TACTIC-E platform trial in the U.K for evaluating the efficacy of EDP1815 in treatment of COVID-19, which has been sponsored by Cambridge University Hospitals NHS Foundation Trust.
The company released positive confirmatory data from a Phase 1b in atopic dermatitis in January 2021 and is awaiting regulatory approval to initiate a Phase 2 trial in 3Q 2021.
Evelo plans to initiate clinical development of EDP2939, a micro microbial extracellular vesicle program in inflammation in 2022. The Company also intends to advance EDP1908, its lead clinical candidate in oncology, which has demonstrated superior tumor growth control and dose-dependent reduction in tumor growth in preclinical studies.
Key Takeaways
Disclosure: I/we have no positions in any stocks mentioned, and no plans to initiate any positions within the next 72 hours.
Click here to please visit our detailed disclosure
References
https://www.marketdataforecast.com/market-reports/atopic-dermatitis-treatment-market
https://ir.evelobio.com/static-files/d70eccdb-39e5-4ee2-b073-82824158f1a9





No Comments