
25 May 5 Stocks that are all set to create waves in the coming days!
While Industries across the globe are grappling with the pandemic and its impact on their operations, Technology Industry has been focused on minimizing the impact and marching ahead with its innovations and breakthroughs, be it electric charging, semiconductor solutions or critical communication systems.
We take a look at some tech companies that are impervious to the scare and offer a very attractive upside in the long run. These companies not only have an excellent potential for growth and returns but have major upcoming catalysts such as new products and services, growing client base or even global expansion plans lined up for 2021.
On the other hand, Biotech Companies continue to show resurgence even in these challenging times, with more and more companies persevering with bringing their clinical candidates to fruition.
We look at some companies that are sure to create waves in the coming days.
Infinera Corp (NASDAQ: INFN)
Market Cap: $ 1.91B; Current Share Price: 9.36 USD
Data by YCharts
The Company is trying to gain a strong foothold in the optical technology market and is competing against the likes of Ciena and Huawei. Optical transmission is rapidly emerging as one of the fastest growing industries owing to reliability and ease of implementation. Wavelength Division multiplexing is emerging as one of the most focal segments of the global optical equipment market.
Infinera recently expanded its portfolio of optical networking solutions that leverage its XR optics technology to offer ICE-XR pluggables for point-to-point and point-to-multipoint transport applications. The Company is likely to benefit from the growing demand for bandwidth and the resistance to Chinese manufacturers amid political tensions. This offers an opportunity for Infinera to fill the gap created by Huawei and the Company is upbeat about its investments in operational improvements bearing fruit this year, providing it with the perfect avenue for growth and expansion.
The technology is extremely popular and much sought after as it allows telecommunication players to expand their network offering, without having to lay down actual fiber. These enable the telecommunication provider to assimilate technological development in their existing optical structure and increase or decrease the capacity of a link, without having to make any changes to the existing network.
According to a report by Acumen Research and Consulting, the global coherent optical equipment market will be worth over 70.14 billion by 2026, growing at a CAGR of 9.23 percent, from 34.61 billion in 2018. This offers a great opportunity for Companies such as Infinera to gain a strong foothold in the market.
Infinera is the creator of the industry’s first point-to-multipoint coherent optical subcarrier aggregation technology. The Company offers diverse solutions encompassing areas such as Intelligent Automation, 5G, Fiber Deep, DAA, cloud networking, business services, access and aggregation, in addition to core and subsea networks.
The Company’s offerings cater to the end-to-end need for networking solutions of Tier 1 carriers, internet content providers, cable operators, government, and enterprise networks worldwide, by using technology innovation. Infinera’s end-to-end, multi-layer packet optical and IP transport solutions facilitate scaling of bandwidth on demand, accelerate service provisioning, and automate operational tasks as per the Company.

Image Source: Company
Infinera’s proprietary vertically integrated Infinite Capacity Engine (ICE) allows the Company to offer economic and robust performance in the long-haul, subsea, data center interconnect, and metro transport applications. The services of the Company range from optical line systems, packet optical transport systems, compact modular platforms, carrier-grade routers, and management and automation solutions.
The Company offers differentiated photonic integrated circuit (PIC) and application-specific integrated circuit (ASIC) chips, which have been developed in-house. Infinera services nine of the top 10 global Tier 1 service providers, the top six ICPs, cable multiple systems operators (MSOs), cloud and data center operators, enterprises and government agencies according to the Company. It offers the lowest Total Cost of Ownership (TCO) and a “pay as you win” instant bandwidth operational model and has a customer base of over 500 clients worldwide, spanning six continents in more than 100 countries. The intellectual property portfolio of Infinera has more than 2000 patents.
Viking Therapeutics (NASDAQ: VKTX)
Market Cap: $ 458.45M; Current Share Price: 5.87 USD
Data by YCharts
A biotechnology company committed to developing therapeutic solutions for endocrine and metabolism related disorder, Viking Therapeutics is currently developing VK2809, a small molecule selective thyroid hormone receptor beta agonist targeted at treatment of non-alcoholic steatohepatitis (NASH), non-alcoholic fatty liver diseases (NAFLD) and other lipid and metabolic disorders. The candidate is currently undergoing a Phase 2b study in biopsy-confirmed non-alcoholic steatohepatitis (NASH) and fibrosis.
According to an estimate only 20 percent of the people suffering from NAFLD have NASH, while the rest only have a simple fatty liver. While nearly 40 percent in the U.S are afflicted with NAFLD, around 3 to 4 percent have NASH. The American Liver foundation estimates that over 100 million people suffer from NAFLD in the U.S alone. According to a report by Reports and Data, the Global NASH market will be worth over USD 13.38 Billion by 2026. The healthcare costs associated with this disease are likely to reach USD 18 billion by 2030 from USD 5 billion now, if the disease is left untreated.
VK2809 has demonstrated the ability to reduce LDL-C and liver fat content in a Phase 2 trial in non-alcoholic fatty liver diseases (NAFLD). The Company is also developing VK0214, an oral small molecule selective thyroid hormone receptor beta agonist, intended for the treatment of X- linked adrenoleukodystrophy(X-ALD).
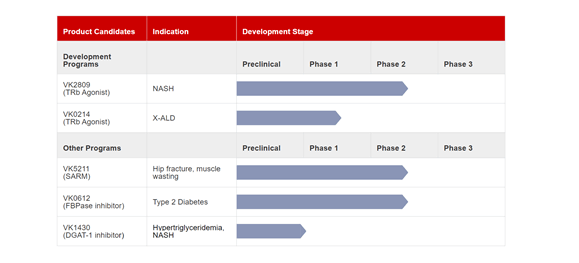
Image Source: Company
The Company is also working on the development of candidates such as VK5211, a non-steroidal selective androgen receptor modulator, which has shown the potential to improve measures of lean body mass in a phase 2 trial in patients recovering from hip fractures. Viking also has two more product candidates in the preclinical stages. The company is in a licensing agreement with Ligand Pharmaceuticals Inc. for use of its small molecules and holds exclusive worldwide rights to a portfolio of five therapeutic drugs.
Viking is advancing VK0612, an oral inhibitor of fructose-1,6 bisphosphatase for treatment of Type 2 diabetes. The candidate has completed six phase 1 trials and a phase 2a proof of concept study and has shown the ability to reduce fasting plasma glucose (fpg) with a favorable safety and efficacy profile. The Company is also developing VK1430 for treatment of Hypertriglyceridemia and NASH.
Provention Bio (NASDAQ: PRVB)
Market Cap: $ 505.73M; Current Share Price: 7.98 USD
Data by YCharts
Provention’s preemptive approach involves developing therapeutic solutions that intercept and prevent before the targeted disease can even take root. The Company’s primary focus is on autoimmune conditions and it actively seeks technologies or targets designed to predict, pre-empt or intervene diseases. Its transformational drug development strategy involves identifying candidates that are underdeveloped due to insufficient clinical trial efficacy or strategic reasons, or those that have demonstrated proof-of-mechanism in intercepting immunopathologic pathways.
In November 2019, the Company announced the grant of a PRIority MEdicines (PRIME) designation by the European Medicines Agency (EMA), for PRV-031 (teplizumab), indicated for the prevention or delay of clinical type 1 diabetes (T1D) in individuals at-risk of developing the disease. PRV-031(teplizumab) is an anti-CD3 monoclonal antibody (mAb), which has undergone multiple clinical studies involving more than 1000 subjects.
The Company is currently engaged in a phase III PROTECT Study, to evaluate the safety and efficacy of PRV-031 in patients newly diagnosed with clinical T1D and expects to complete the enrolment in H2,2021. The drug candidate has consistently demonstrated an ability to preserve beta cell function and lower the need for exogenous insulin usage in previous clinical studies. The Company acquired the candidate from MacroGenics.
In January 2021, The Company filed a Biologics License Application (BLA) for teplizumab for the delay or prevention of clinical type 1 diabetes (T1D) in at-risk individuals and was granted a Priority Review and assigned a user fee goal date of July 2, 2021, under the Prescription Drug User-Fee Act (PDUFA) by the U.S. Food and Drug Administration (FDA).
According to the International Diabetes Federation, the number of people living with diabetes will rise to 629 million by 2045, from 425 million adults (20-79 years) in 2017. In the U.S alone the people living with diabetes are set to increase from 58 million in 2017 to 67 million by 2045.
The increased awareness about the long-term complications caused by diabetes, along with the rise in co-morbidities such as cardiovascular, renal and neuropathic complications is set to propel the growth in the segment from $26.8 billion in 2016 to around $64 billion by 2026 according to a research by GlobalData.
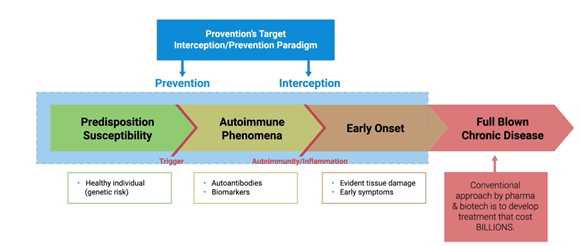
Image Source: Company
The company’s lead candidate for the treatment of Crohn’s disease, PRV-6527, as an oral Colony Stimulating Factor-1 Receptor (CSF-1R) small molecule inhibitor, which achieved full patient enrolment for its Phase 2a PRINCE (PRovention Investigation in Crohn’s DiseasE) clinical trial. PRV-6527 was deemed to have the potential to intercept the differentiation of inflammatory dendritic cells and macrophages and inhibit their action in the intestinal mucosa in Crohn’s disease. It has already been proven to inhibit CSF-1R signaling and myeloid cell counts in blood in PoC studies while being developed by Janssen. The Company faced a setback in its Phase 2a clinical trial, PRINCE, evaluating the PRV-6527 for the treatment of Crohn’s disease, with patients failing to respond adequately and the trial not meeting its primary endpoint of a statistically significant change in a scale called CDAI.

Image Source: Company
In addition, Prevention is also developing PRV-015, an anti-IL-15mAb for Gluten dependent celiac disease in partnership with Amgen and is expecting the top-line results from a phase 2b trial in H2,2021, Furthermore, the Company is also advancing PRV-3279, a B-Cell autoimmunity checkpoint, which is being evaluated for the treatment of systemic lupus erythematosus and prevention of immunogenicity. The Company intends to initiate a phase 2a trial in lupus in H2,2021.
Market Cap: $ 494.03M; Current Share Price: 5.00 USD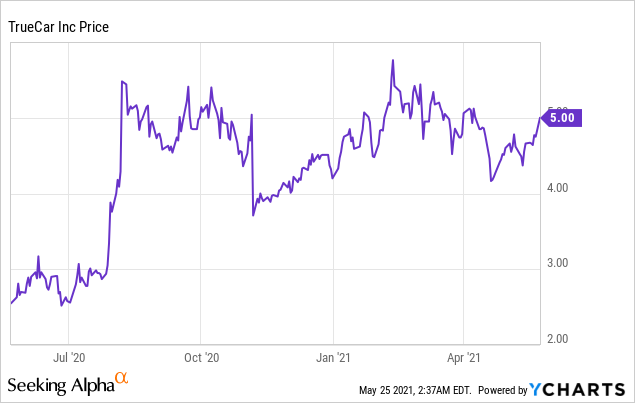
Data by YCharts
TrueCar is a digital automotive marketplace that enables users to make informed buying choices while purchasing new or used cars from dealers across the country. The Company has established a robust network of over 14000 certified dealers and offers car-buying programs to over 250 leading brands including Sam’s Club, and American Express.
In May 2021, the Company announced the launch of a pilot program in association with Roadster Inc, which will enable TrueCar users to finalize their car buying deals digitally through Roadster’s back-end software with the help of an API. The facility is currently available through more than 650 dealers that work with both the companies.
The Company’s website boasts of over 8 million unique monthly users and has successfully sold more than 750,000 units till date. As per its Q1,2021 financial results, TrueCar reported a total revenue of $65.1 million, an increase of 1.8% as compared to Q4,2020. In addition, there were 9.2 million average monthly unique visitors in Q1,2021 compared to 7.8 million in Q1,2020. However, the number of units sold went down by 15.8% year-over-year and stood at 165,858 in Q1,2021. Monetization was reported to be $391 in the first quarter of 2021 and was the same as last year. TrueCar expects the revenues to be in the range of $65 million to $66 million for the quarter ending June 30, 2021, while the Adjusted EBITDA is expected to be above breakeven.
The Car Industry, which was impacted by the COVID-19 pandemic and witnessed lower purchasing, is now bouncing back to normalcy with the economies around the world opening up. However, job losses, lower discretionary incomes and increasing unemployment may prove a deterrent to the growth of the Company.
The Company provides a valuable service by redesigning the way customers evaluate and buy cars by connecting them with potential dealers offering lucrative deals online, for a nominal fee. This particular market has very low barriers to entry and the Company has many competitors, each trying to find a better way to connect potential customers and dealers. TrueCar therefore has its task set out in front of it, which is to find ways and means to not only drive user retention but also fend off potential competitors, if it intends to stay one step ahead of competition.
Market Cap: $ 27.30M; Current Share Price: 4.20 USD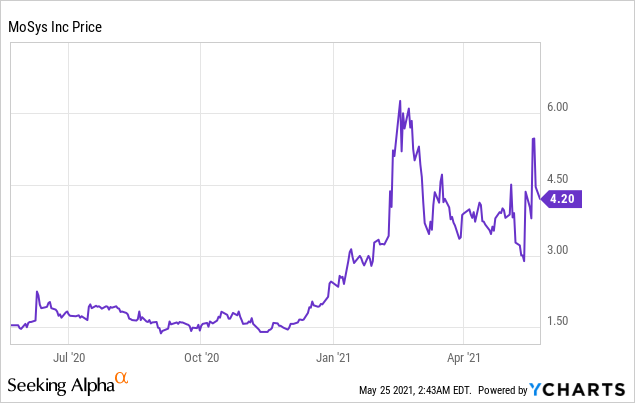
Data by YCharts
MoSys is a semiconductor manufacturer that develops MoSys® Bandwidth Engine integrated circuits (ICs) for companies engaged in real-time bandwidth-intensive services such as video-on-demand, IP TV and cloud computing. The Company’s IC solutions remove bottlenecks in critical data throughput and deliver speeds and intelligence for systems with aggregate rates above 100 Gigabits per second (Gbps), thereby improving their performance and delivering economies.
The Company is an ISO 9000 certified manufacturer and is qualified to meet carrier-grade reliability requirements. MoSys family of IC’s, Programmable HyperSpeed Engine (PHE) ICs, and LineSpeed™ 100G PHY products leverage proven serial interface technology and high-density integrated memory. MoSys sells various products under the brand names Quazar, Blazar and Stellar.

Image Source: Company
In April,2021, the Company expanded its patent portfolio by acquiring five patents related to search and classification algorithms. MoSys’s Stellar Packet Classification IP, which is based on the Company’s proprietary Graph Memory Engine (GME) technology, uses these algorithms to provide differentiated networking search or data processing solutions for Cloud infrastructure applications. In addition, it will help OEMs and network operators offer configurable platforms which can support millions of rules at hundreds of gigabits per second.
The U.S Senate unveiled bipartisan legislation, which calls for a $52 billion investment to improve research and investment in semiconductor chip production over the next five years. The proposal will be part of a 1,400-page revised bill that seeks to invest $120 billion on technology research. According to a report by Reuters, the bill includes $39 billion in production and R&D incentives and $10.5 billion to implement programs including the National Semiconductor Technology Center, National Advanced Packaging Manufacturing Program and other R&D programs. The bill comes amidst growing concerns about growing Chinese dominance in this key industry and the need to find viable alternatives to Huawei and ZTE Corp.
Companies such as MoSys will be able to benefit from the legislation as there is a greater push on indigenous semiconductor manufacturing.
Disclosure: I/we have no positions in any stocks mentioned, and no plans to initiate any positions within the next 72 hours.
Click here to please visit our detailed disclosure
References
https://www.vikingtherapeutics.com/
https://www.niddk.nih.gov/health-information/liver-disease/nafld-nash/definition-facts
https://proventionbio.com/intercepting-autoimmune-disease
https://ir.truecar.com/2021-05-06-TrueCar-Reports-First-Quarter-2021-Financial-Results
https://finance.yahoo.com/news/mosys-stock-trading-higher-acquiring-123451760.html
https://investor.mosys.com/MOSY





No Comments